All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The lym Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the lym Hub cannot guarantee the accuracy of translated content. The lym and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
AACR 2017 | Poster 154/21 – Copanlisib is active as monotherapy or in combination in Mantle Cell Lymphoma, Marginal Zone Lymphoma, and Chronic Lymphocytic Leukemia cell lines
This year’s American Association for Cancer Research (AACR) annual meeting took place on 1–5 April in Washington, DC, USA. The program committee Chair was Kornelia Polyak, MD, PhD, from the Dana-Farber Cancer Institute, Boston, Massachusetts.
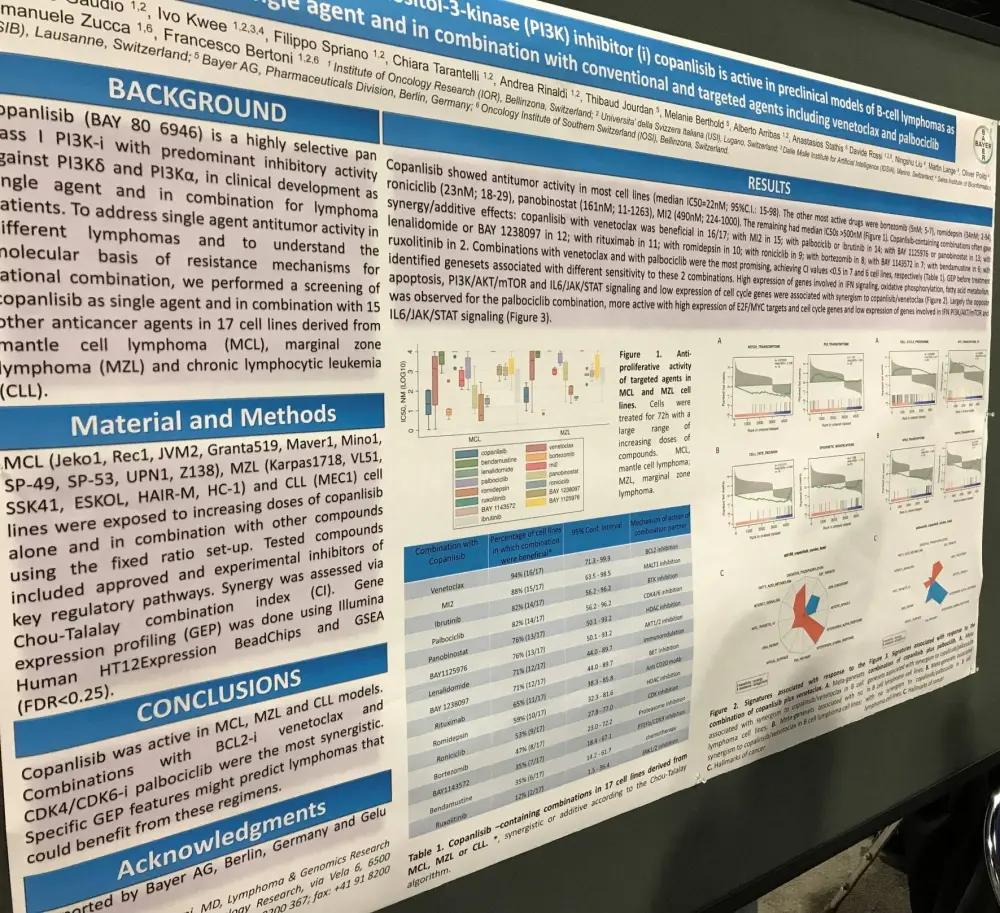
On Sunday 2nd April, a poster (154 / 21) by Eugenio Gaudio, from the Institute of Oncology Research, Bellinzona, Switzerland, et al. titled “The phosphatidylinositol-3-kinase (PI3K) inhibitor (i) copanlisib is active in preclinical models of B-cell lymphomas as single agent and in combination with conventional and targeted agents including venetoclax and palbociclib” was presented.
Multiple MCL, MZL, and CLL cell lines were exposed to increasing doses of copanlisib alone and in combination with other compounds using a fixed ratio set-up.
Key Highlights:
- Copanlisib showed anti-tumor activity in the majority of cell lines (median IC50 = 22nM; 95% CI, 15–98)
- The other most efficacious drugs = bortezomib (5nM; 5–7), romidepsin (34nM; 2–94), roniciclib (23nM; 18–29), panobinostat (161nM; 11–1263), and MI2 (490nM; 224–1,000)
- The remaining had median IC50s >500nM
- Combinations with venetoclax and palbociclib were the most promising, achieving CI values <0.5 in 7 and 6 cell lines, respectively
- Gene expression profiling before treatment identified gene sets associated with sensitivity to these two combinations
- Low expression of cell cycle genes as well as high expression of genes involved in interferon signaling, oxidative phosphorylation, fatty acid metabolism, apoptosis, PI3K/AKT/mTOR, and IL6/JAK/STAT signaling were associated with synergism to copanlisib/venetoclax
- The palbociclib combination was more active with high expression of E2F/MYC targets and cell cycle genes and low expression of genes involved in IFN PI3K/AKT/mTOR and IL6/JAK/STAT signaling
The poster concluded by stating that copanlisib is active in MCL, MZL, and CLL cell lines. Combinations with venetoclax (BCL-2 inhibitor) and palbociclib (CDK4/CDK6 inhibitor) were the most synergistic. Using gene expression profiling may identify patients with Lymphoma who would respond well to these combination regimens.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average time to secure a reimbursed CAR T-cell therapy manufacturing slot for patients with DLBCL (from decision to treatment with a CAR T-cell therapy)?

