All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi and Thermo Fisher Scientific and supported through independent educational grants from Bristol Myers Squibb, Incyte, Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
ASH 2016 | Brentuximab Vedotin Demonstrates Significantly Superior Clinical Outcomes in Patients with CD30-Expressing Cutaneous T Cell Lymphoma Versus Physician's Choice (Methotrexate or Bexarotene): The Phase 3 ALCANZA Study
The 58th Annual Meeting & Exposition of the American Society of Hematology’s (ASH) took place in San Diego, CA, and on December 3rd, Youn H. Kim, MD, from Stanford University School of Medicine, CA, USA, presented the results of the randomized, open-label, Phase-III ALCANZA study into brentuximab vedotin. The study recruited patients with CD-30 positive CTCL.
Highlights:
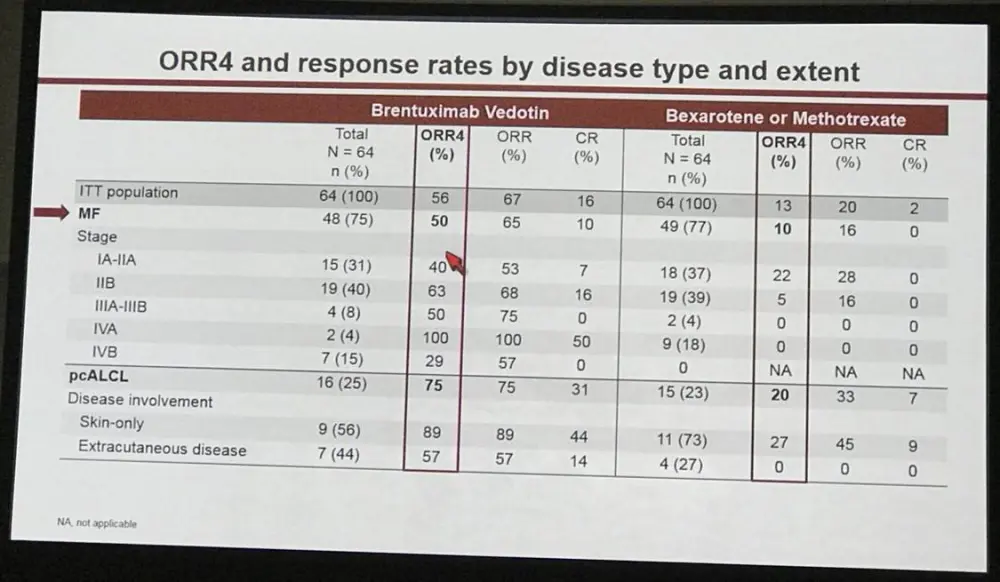
- 131 pts, 128 ITT population (97 mycosis fungoides, 31 pcALCL)
- 64 given BV (1.8mg/kg IV, once every 3 weeks), 64 given physicians choice (PC) of either MTX (5-50mg PO weekly) or Bex (300mg/m2 PO daily) for up to 16x 3week cycles
- Results at median follow-up 17.5 months
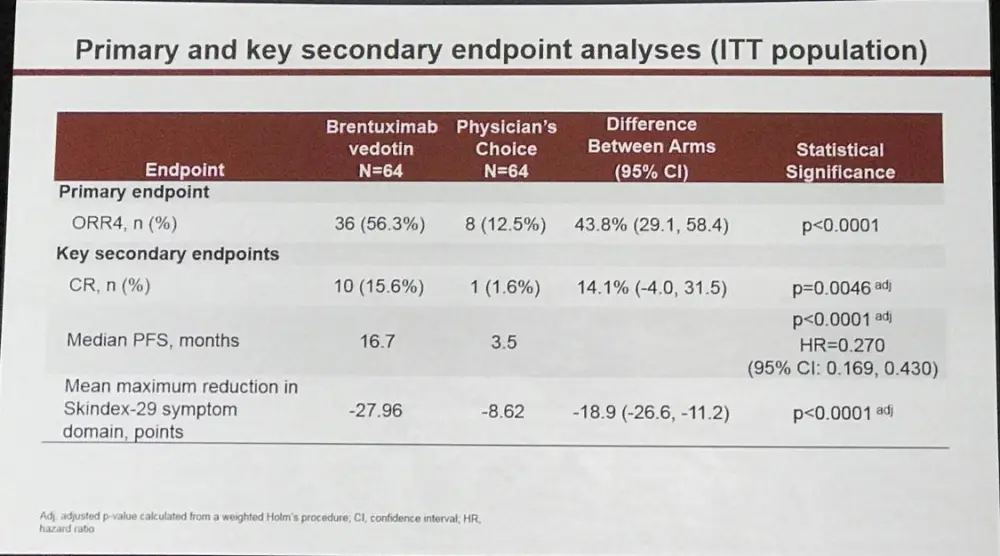
- ORR lasting ≥ 4 months = BV 56% vs PC 13% (P<0.0001)
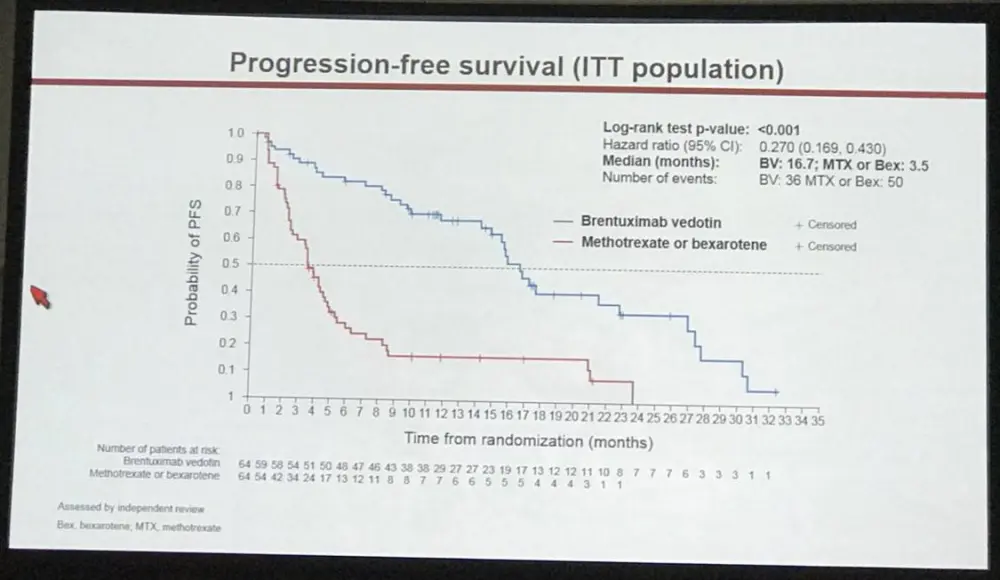
- All AE grade 3-4: BV = 41% vs PC = 47%
- Drug related AE grade 3-4: BV = 29% vs PC = 29%
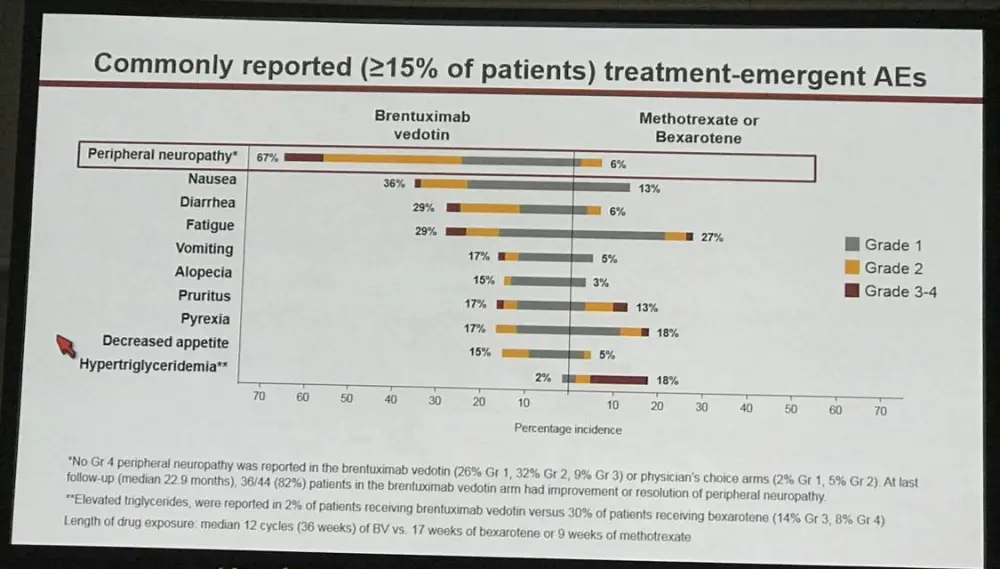
- Peripheral neuropathy: BV = 67% vs PC = 6%. - 82% BV patients had improvement/resolution

Youn H. Kim concluded by stating that BV had a much superior clinical outcome compared with PC of MTX or Bex in CTCL patients and that the data here showed strong evidenced in favor of BV over MTX or Bex in CD-30 positive CTCL patients.

Abstract:
Background: Cutaneous T cell lymphoma (CTCL) is a chronic disease that negatively impacts quality of life (QoL) and, in advanced stages, has poor prognosis. Current systemic therapies rarely provide reliable and durable responses, and to date, no systemic agent has shown outcomes superior to standard-of-care therapy such as methotrexate (MTX) or bexarotene (Bex). CD30-directed antibody-drug conjugate brentuximab vedotin (BV) has demonstrated marked clinical activity in two phase 2 single-arm trials in CTCL with overall response rates (ORR) of about 70%. These results led to ALCANZA, a randomized, open-label, multicenter phase 3 trial of the efficacy and safety of BV vs physician's choice (PC) of MTX or Bex, in previously treated patients with CD30-expressing CTCL (NCT01578499). This is the first reported randomized phase 3 trial testing a new agent against standard-of-care therapy in CTCL.
Methods: Adults with CD30-expressing (≥10% of infiltrate by central review) mycosis fungoides (MF) who received ≥1 prior systemic therapy or primary cutaneous anaplastic large cell lymphoma (pcALCL) who received ≥1 prior systemic therapy or radiotherapy were enrolled. Patients were stratified by diagnosis and randomized 1:1 to receive BV 1.8 mg/kg IV, once every 3 weeks, or PC of either MTX 5–50 mg PO, once weekly, or Bex 300 mg/m² (target dose) PO, once daily, for up to 16 three-week cycles, until disease progression or unacceptable toxicity. Primary endpoint was ORR4, defined as ORR lasting ≥4 months, an endpoint that captures response rate and duration as a single measurement. ORR4 was determined by independent review of global response using the ISCL/EORTC consensus guidelines. Global response is a composite of skin evaluation (modified severity weighted assessment tool), radiographic assessment, and Sézary cell enumeration. Key secondary endpoints were CR rate, PFS, and symptom burden measured by the symptom domain of the Skindex-29 QoL tool. Sample size was calculated to provide 90% power to detect a 30% improvement in ORR4. Treatment-emergent adverse events (AEs) were evaluated according to NCI CTCAE v4.03.
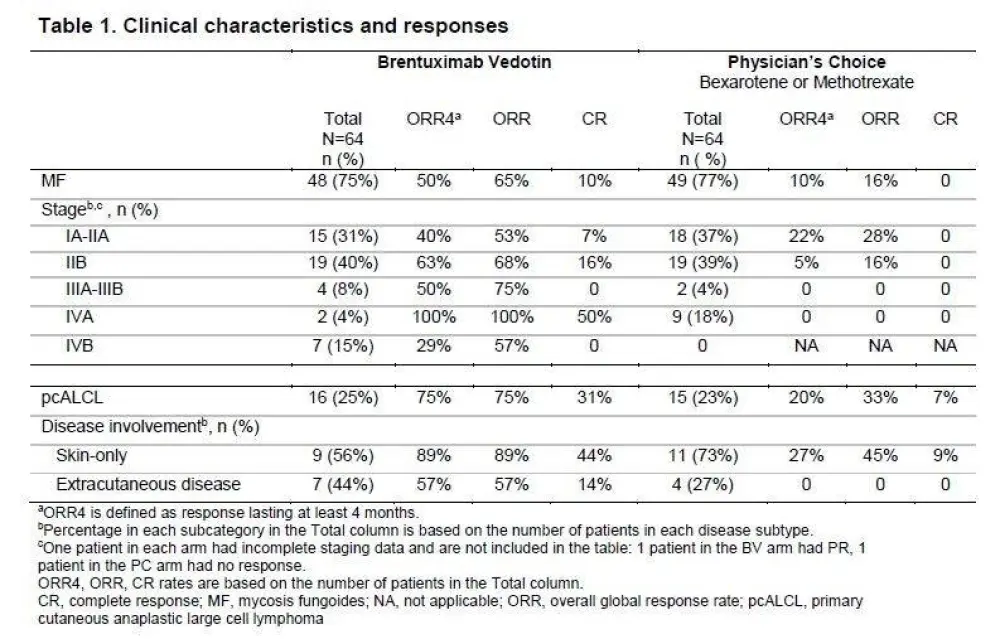
Results: 131 patients were randomized with 128 patients in the intent-to-treat population (97 MF, 31 pcALCL; 3 excluded for insufficient CD30 expression) and assigned to BV (n=64) or PC (n=64). Baseline characteristics were generally balanced between arms with the exception of more patients with extracutaneous disease in the BV arm (Table). In BV vs PC arms, respectively, median age was 62 (22–83) vs 58 (22–83) years; ECOG performance status 0–1 was 95% vs 97%. Patients in each arm had a median of 2 prior systemic therapies.
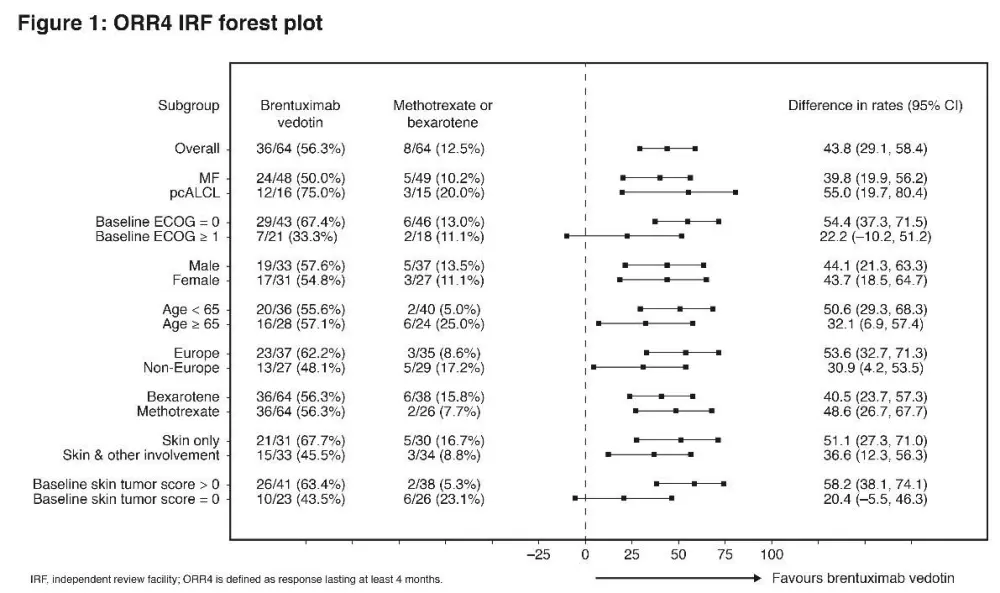
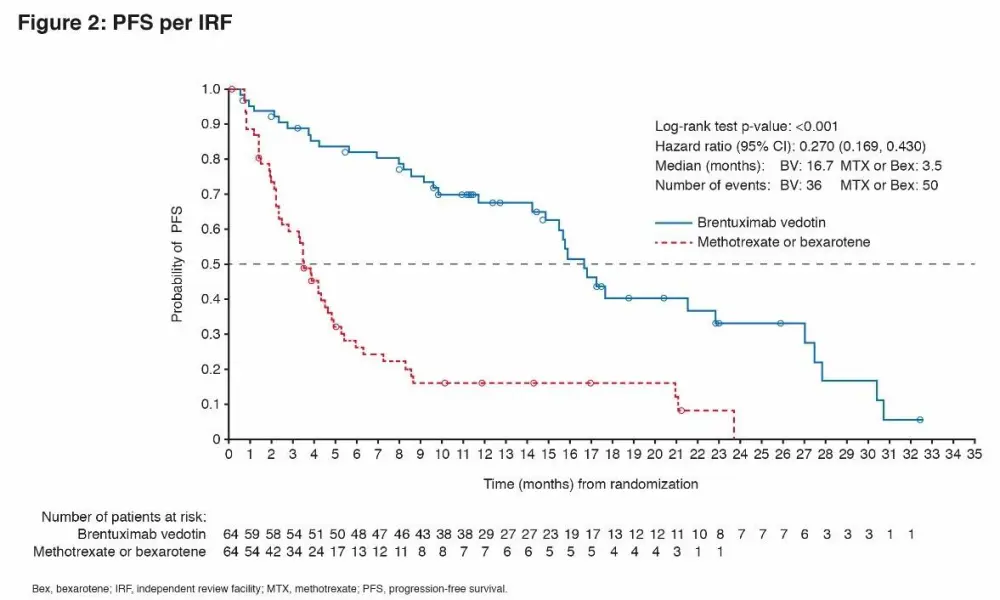
At a median follow-up of 17.5 months, ORR4 (primary endpoint) and PFS strongly favored BV vs PC with ORR4 of 56% vs 13% (p<0.0001) and median PFS of 16.7 vs 3.5 months (HR 0.270; 95% CI, 0.169–0.430; p<0.0001), respectively (Fig 1, 2). ORR was 67% (n=43) with CR 16% for BV, compared with ORR of 20% (n=13) with CR 2% for PC (ORR, p<0.0001; CR, p=0.0046). Skindex-29 showed significantly greater symptom reduction for BV compared with PC (-27.96 vs -8.62; p<0.0001).
Patients received a median of 12 (1–16) cycles of BV vs 5.5 (1–16) Bex and 3 (1–16) cycles of MTX. Grade 3–4 AEs, all cause / drug-related, were seen in 41% / 29% with BV vs 47% / 29% with PC. Serious AEs were seen in 29% of patients in each arm. Peripheral neuropathy (PN), any grade, was seen in 67% in the BV arm (32% grade 2, 9% grade 3) and 6% in the PC arm. At last follow-up, 36/44 (82%) patients in the BV arm had improvement or resolution of PN. Discontinuation due to AEs occurred in 24% (BV) vs 8% (PC). Four deaths in the BV arm (3 unrelated to study drug) occurred within 30 days of the last dose. Other AEs were consistent with reported safety profiles for the individual agents.
Conclusions: In this first report of a randomized phase 3 trial evaluating a new agent vs standard-of-care options in CTCL, the clinical outcome of BV was far superior to PC (MTX or Bex). Highly statistically significant improvements in ORR4 (44%) and median PFS (13.2 months) were observed with BV. Greater ORR with higher CR rate and reduction in symptoms per Skindex-29 symptom domain were also observed in the BV arm. Safety data for BV were consistent with the established tolerability profile. The data from this randomized trial provide compelling evidence favoring BV over PC of Bex or MTX in CD30-expressing CTCL.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

