All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
ASH 2016 | Ibrutinib in combination with lenalidomide and rituximab in R/R DLBCL
The 58th Annual Meeting & Exposition of the American Society of Hematology (ASH) took place in San Diego, CA, on December 3–6, 2016. On Sunday 4th December, an oral abstract session was held between 4:30pm and 6:00pm in the “Aggressive Lymphoma (Diffuse Large B-Cell and other aggressive B-Cell Non-Hodgkin Lymphomas) – results from prospective clinical trials: Novel Approaches in Aggressive Lymphoma” category. This session was moderated by Jason R. Westin, MD MS, from The University of Texas MD Anderson Cancer Center, and Laurie H Sehn, MD, of the British Columbia Cancer Agency.
Abstract #473 was presented during this session, titled “A multicenter open-label, phase 1b/2 study of ibrutinib in combination with lenalidomide and rituximab in patients with Relapsed or Refractory (R/R) Diffuse Large B- Cell Lymphoma (DLBCL)” by Andre Goy, MD, of the John Theurer Cancer Center, NJ, and colleagues.
This group conducted an open-label multicenter phase Ib/II study (NCT02077166) designed to assess the safety and efficacy of ibrutinib in combination with lenalidomide and rituximab in patients with R/R DLBCL. Determining the Maximum Tolerated Dose (MTD) and/or Recommended Phase II (RP2) dose of lenalidomide in combination with fixed doses of ibrutinib and rituximab were the primary objectives for the phase Ib study. Overall Response Rate (ORR) was the primary objective for the phase II study.
Patients received 560mg once daily of ibrutinib in combination with intravenous rituximab (375mg/m2) and an escalating dose of lenalidomide. The doses of lenalidomide used are as follow: 15mg starting dose (Cohort 1), 10mg de-escalation dose (Cohort -1), 15mg re-escalation (Cohort +1) and 20mg (Cohort 2). Thirty-seven patients were treated across all cohorts in this trial; 20 non-GCB/ABC (18 non-GCB, 2 ABC), 9 GCB, 8 unknown DLBCL subtype; median age was 63 years (41-85).
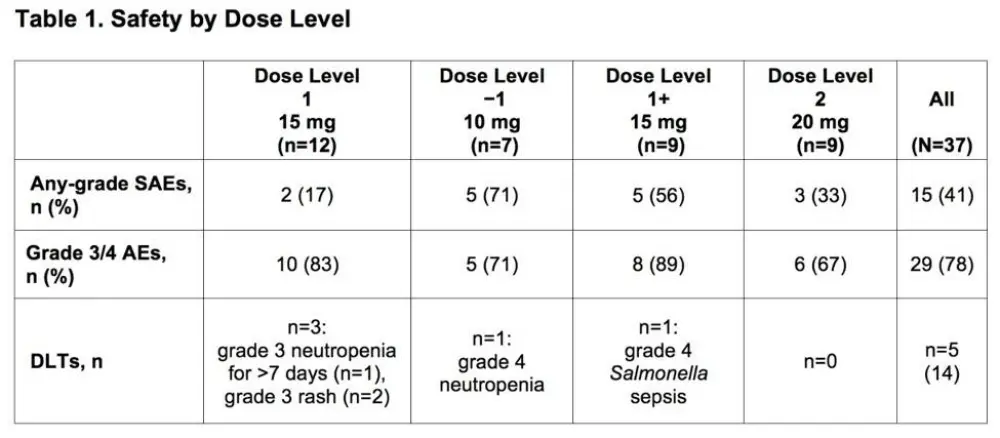
- Grade 3/4 AEs occurred in 29 patients (Table 1)
- Frequent grade 3/4 AEs include neutropenia (32%), thrombocytopenia (14%) and maculopapular rash (11%)
- Fifteen patients had any-grade Serious AEs (SAEs) (Table 1)
- Five Patients had Dose-Limiting Toxicities (DLTs) (Table 1)
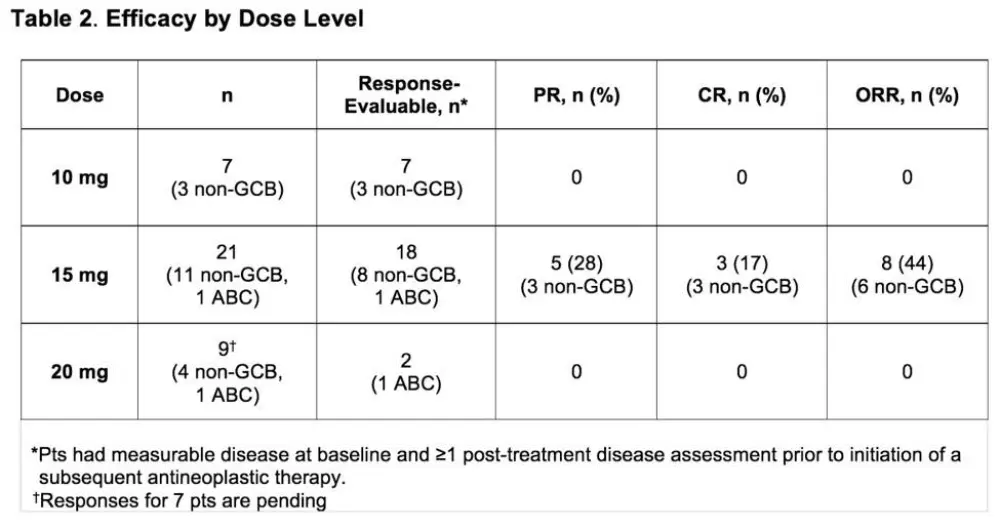
- In the response-evaluable population; no response (10mg, n = 7), 44% ORR, 19% Complete Responses (CR), 31% Partial Responses (PR) (15mg, n = 16), 13% PR (20mg, n = 8)
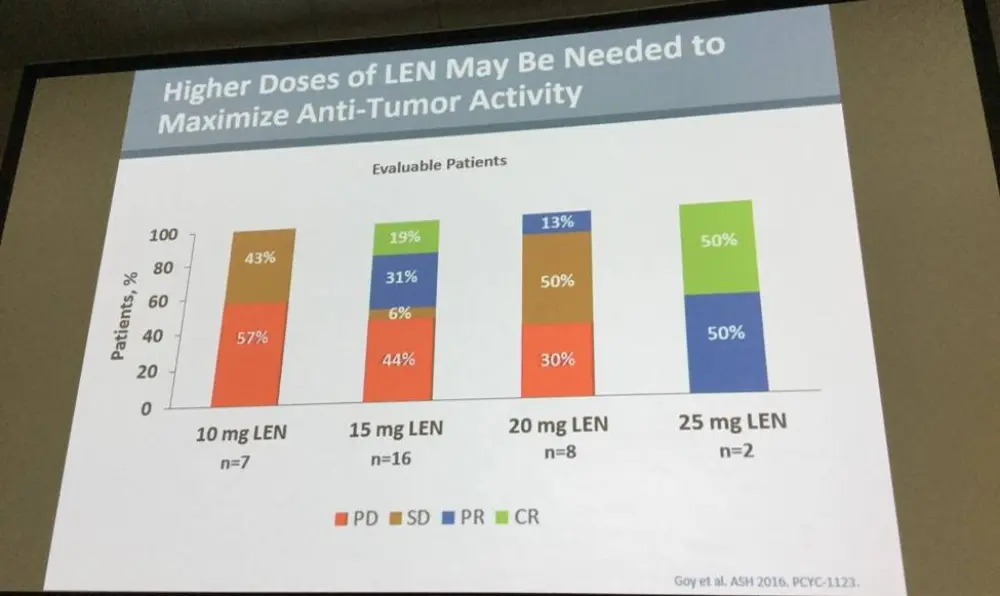
The data from the phase Ib study suggests 20mg lenalidomide dose level combined with rituximab and ibrutinib should be used in the phase II portion of this study. Preliminary results obtained show responses at 15mg lenalidomide, thus indicating that a dose level above 10mg lenalidomide might be required to induce responses in DLBCL. The group concluded their presentation by suggesting that, despite the small amount of patients in this trial, the data obtained in the high-risk DLBCL patients were encouraging.

Abstract
Background: DLBCL is the most common subtype (30-40%) of non-Hodgkin lymphoma and 50-60% of patients (pts) are cured with standard R-CHOP therapy. Among pts who fail R-CHOP, only 10-20% experience prolonged disease-free intervals with high-dose chemotherapy and autologous stem cell transplant (SCT). In particular, pts relapsing within 12 months (mo) of diagnosis and/or refractory pts have a very poor outcome (Crump, ASCO 2016). In addition, many pts are SCT–ineligible due to advanced age and/or comorbidities (Friedberg, ASH Educ Program 2011). Therefore, R/R DLBCL represents a clear unmet medical need. Ibrutinib (ibr), a first-in-class, oral, once-daily inhibitor of Bruton’s tyrosine kinase, has shown activity as a single agent in R/R DLBCL, particularly in the non-germinal center B-cell–like (non-GCB) subtype (Wilson, Nat Med 2015). Lenalidomide (LEN) is an immunomodulatory agent that has shown activity in combination with rituximab (RTX) in phase 2 studies of R/R DLBCL (Wang, Leukemia 2013). This multicenter, open-label, phase 1b/2 study is designed to evaluate the safety and efficacy of ibr in combination with LEN and RTX in pts with R/R DLBCL. Here, we report preliminary results of the phase 1b portion of the study.
Methods: The primary objective of the phase 1b portion is to determine the maximum tolerated dose and/or recommended phase 2 dose of ibr in combination with LEN and RTX in R/R DLBCL using a modified 3+3+3 design; the secondary objective is overall response rate (ORR) (Cheson, J Clin Oncol 2007). In phase 2, efficacy of the combination will be evaluated in R/R non-GCB DLBCL. SCT-ineligible pts, aged ≥18 years with histologically confirmed R/R DLBCL after ≥1 prior therapy, ECOG PS <2, and no central nervous system disease or history of allogeneic SCT were included. Pts received once-daily ibr (560 mg) in combination with IV RTX 375 mg/m2 on Day (D)1 of each 28-day cycle for 6 cycles and escalating doses of LEN on D1-21 of each 28-day cycle: 15 mg starting dose = Dose Level 1 (Cohort 1), 10 mg de-escalation cohort = Dose Level −1 (Cohort −1), 15 mg re-escalation cohort = Dose Level 1 (Cohort 1+), and 20 mg Dose Level 2 (Cohort 2). Dose Level Review Committee (DLRC) meetings were held for each dose level.
Results: Thirty-seven pts were treated across all cohorts; 20/37 had non-GCB/ABC (18 non-GCB, 2 ABC), 9/37 GCB, and 8/37 unknown DLBCL subtype;10/37 transformed disease (2 GCB, 3 non-GCB, 1 ABC, 4 unknown). Median age was 63 years (range 41-85); 16 pts (43%) were ≥65 years old. Median time from diagnosis was 14.3 mo. Pts had a median of 3 prior regimens (range, 1-5); 23 (62%) were considered refractory to their last regimen. Grade 3/4 adverse events (AEs) occurred in 29 (78%) pts. The most frequent (>10%) grade 3/4 AEs were neutropenia (32%), thrombocytopenia (14%) and maculopapular rash (11%). Any-grade serious AEs (SAEs) occurred in 15 (41%) pts (Table 1). Dose-limiting toxicities (DLTs) were observed in 5 (13%) pts (rash [2], neutropenia [2], sepsis [1]; Table 1). Due to 3 DLTs in Cohort 1, a cohort at Dose Level −1 was opened. As 1 DLT was observed at this dose level, re-escalation to higher levels (starting at 15 mg LEN) occurred. Twenty-four (65%) pts discontinued treatment due to disease progression (14/24), AE (8/24), and withdrawal of consent (2/24); 14 (38%) died, primarily due to progressive disease (n=9). Deaths due to AEs (n=4) included E. coli sepsis, cardiac arrest (after progression and subsequent antineoplastic therapy), pneumonia, and worsening DLBCL. Cause of death was unknown in 1 pt. In the response-evaluable population (Table 2), on 10 mg LEN (n=7), there were no responses. On 15 mg LEN (n=18, including 2 pts with DLTs who received ≤15 days of study drug), the ORR was 44%; there were 3 complete responses (CRs) and 5 partial responses (PRs, among them, 3 pts with refractory disease); 6/9 (67%) with non-GCB/ABC DLBCL responded (including all 3 CRs). On 20 mg LEN, 2 response-evaluable pts did not respond, other response evaluations are pending.
Conclusions: Based on safety data from the phase 1b portion of the study and the DLRC recommendation, the phase 2 portion of the study is being initiated at the 20 mg LEN dose level in combination with RTX and ibr. Preliminary efficacy results demonstrate responses at 15 mg LEN and suggest that a dose level above 10 mg LEN might be needed to induce a response in R/R DLBCL. In summary, despite small pt numbers, the results seen in this high-risk refractory DLBCL population are encouraging.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average time to secure a reimbursed CAR T-cell therapy manufacturing slot for patients with DLBCL (from decision to treatment with a CAR T-cell therapy)?

