All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi and Thermo Fisher Scientific and supported through independent educational grants from Bristol Myers Squibb, Incyte, Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
ASH 2016 | Primary Results of the GALLIUM Study
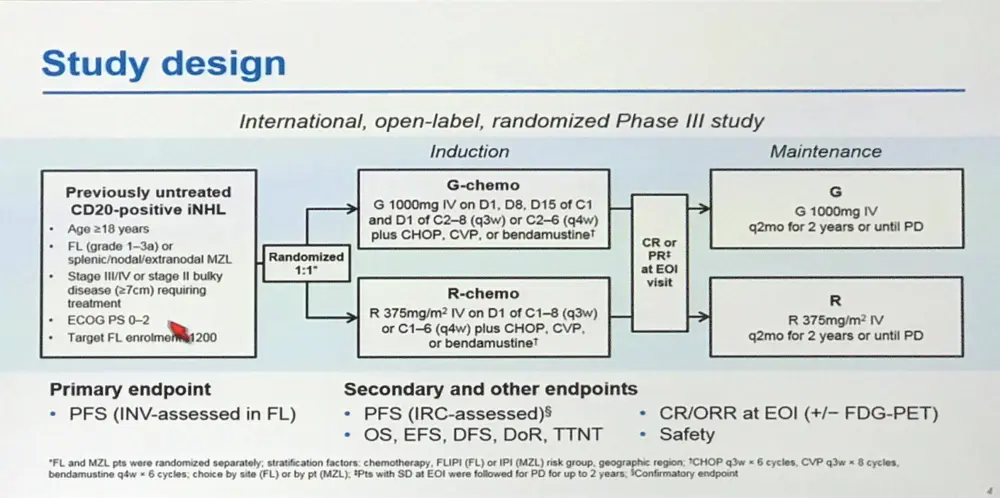
The 58th Annual Meeting & Exposition of the American Society of Hematology (ASH) took place in San Diego, CA, and on December 4th, Robert E. Marcus, MBBS, FRCP, FRCPath, from Kings College Hospital, London, presented the primary results for patients with FL as part of the randomized Phase-III GALLIUM study. The GALLIUM study is a global, multi-center, open-label study comparing the safety profile and efficacy of front-line obinutuzumab (G) or rituximab (R) in combination with chemotherapy in previously untreated Follicular Lymphoma (FL) patients.

Upon introducing the presentation, John P. Leonard, MD, from Weill Cornell Medicine, New York, raised important questions to consider when digesting the GALLIUM results. Key among these were that PFS does not necessarily provide sufficient data about the patients’ quality of life or clinical benefit and that OS remains the ultimate measure for efficacy. Additionally, many patients refuse maintenance therapies for different reasons, therefore how applicable would these results be for this population?

Highlights:
- 1202 FL pts enrolled and randomized 1:1 = 601 G-chemo, 601 R-chemo
- Chemotherapy options: Bendamustine (n=827), CHOP (n=433) or CVP (n=141)
- Baseline pts characteristics: R-chemo group had slightly more patients with high FLIPI scores and bulky disease (≥7cm)
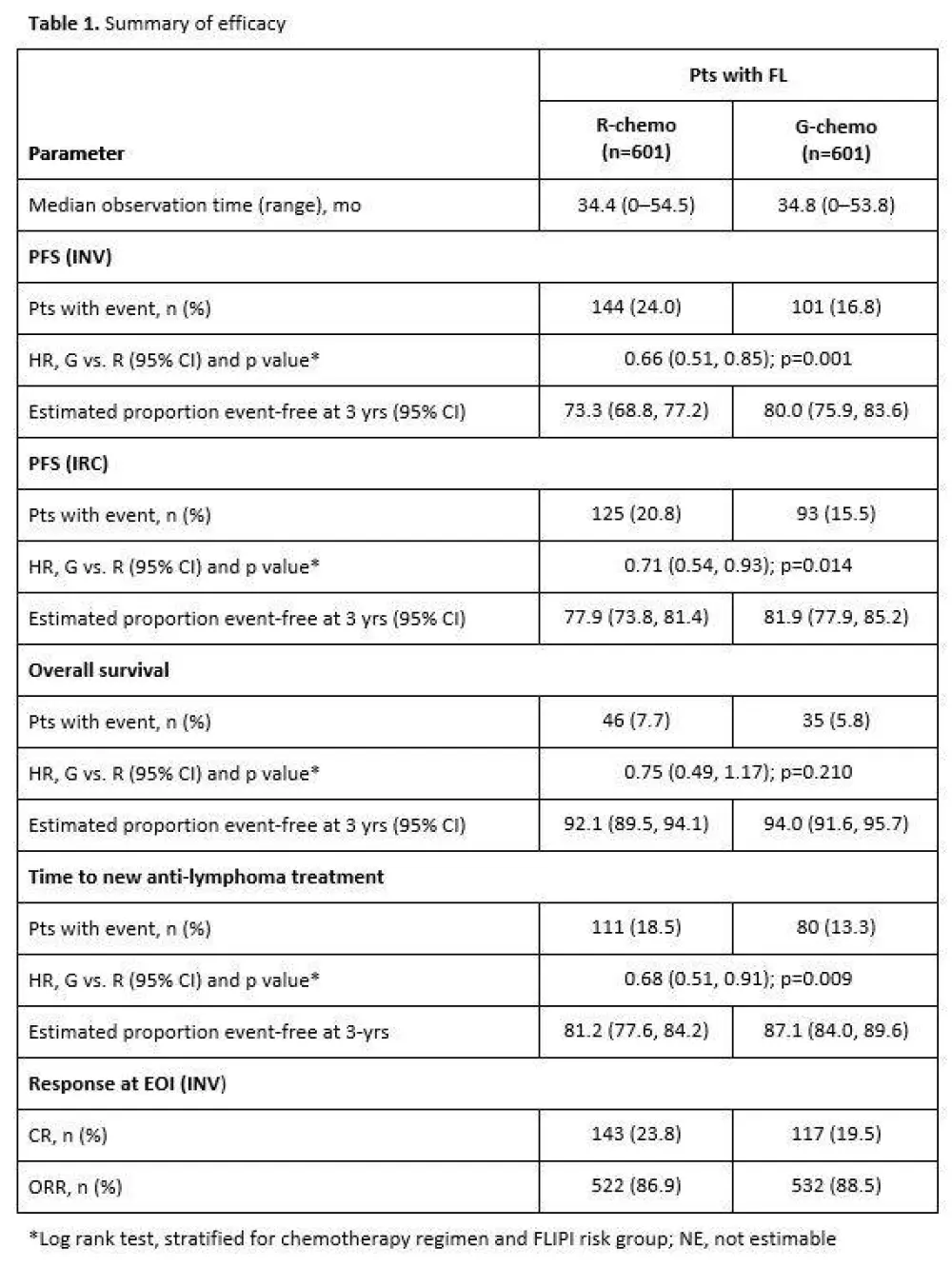
- Results at median follow-up 34.5 months
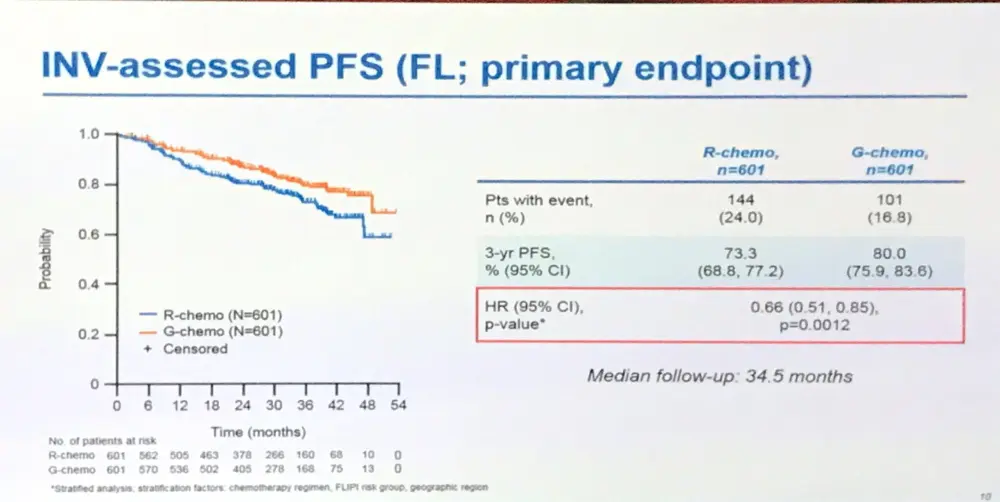
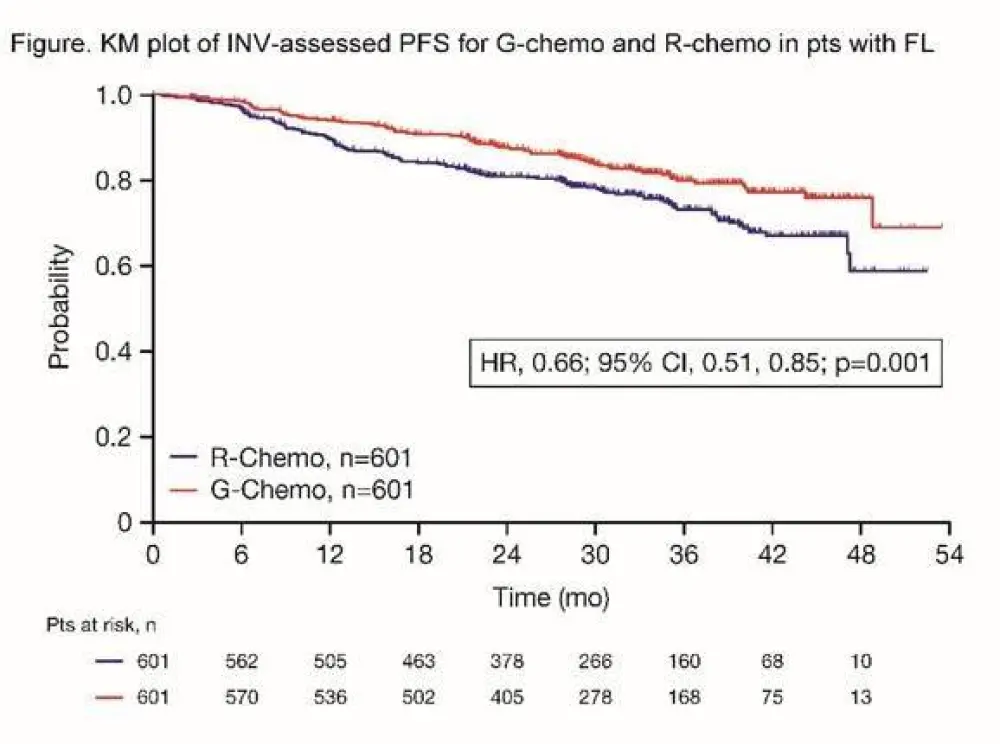
- Investigator assessed 3-year PFS: R = 73.3%, G = 80.0%, HR = 0.66 (p=0.0012)
- Review committee assessed 3-year PFS: R = 77.9%, G = 81.9%, HR = 0.71 (p=0.0138)
- 3-year OS: R = 92.1%, G = 94%, HR = 0.75 (p=0.21)
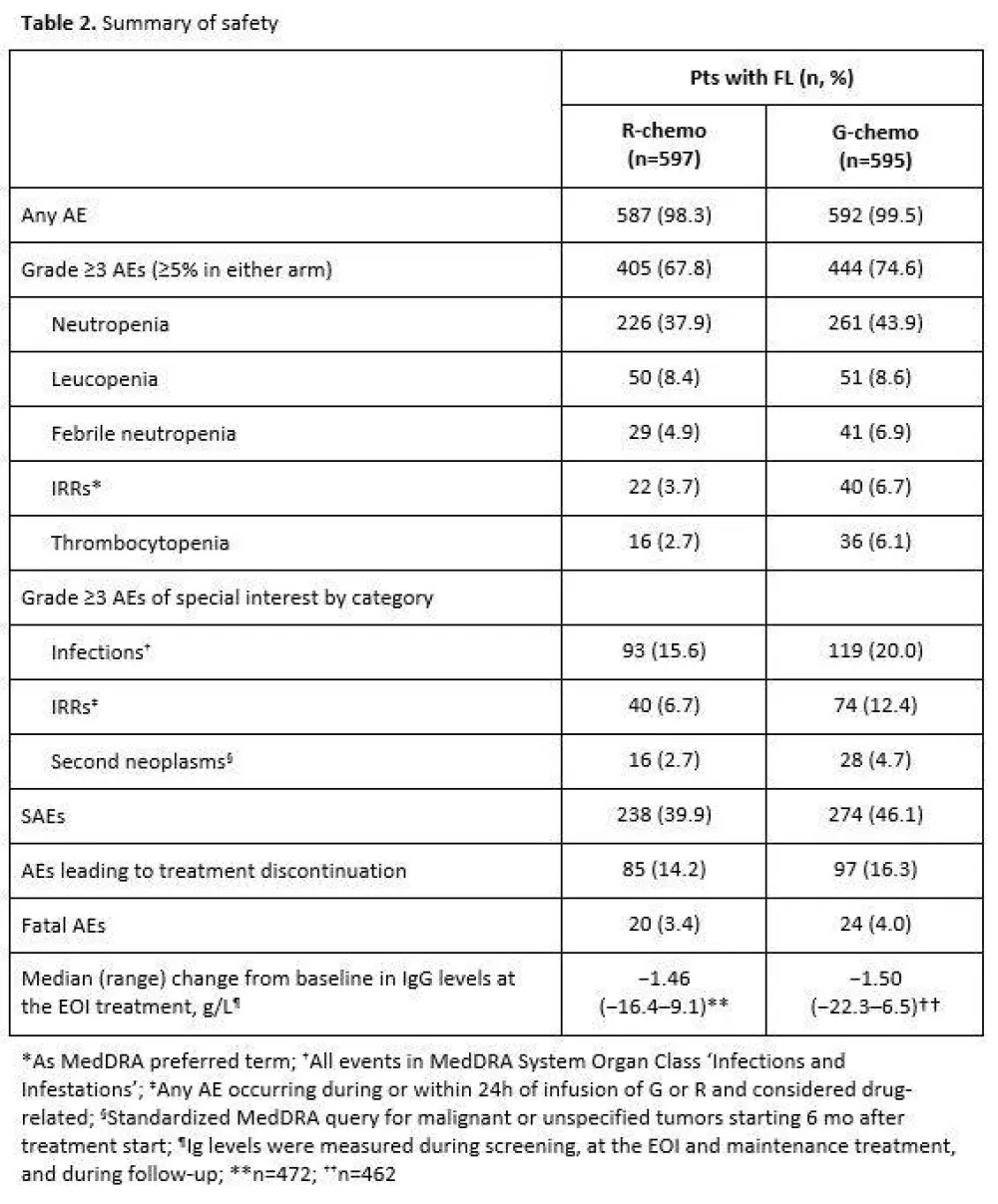
- Adverse events:
- ≥ Grade 3 AEs: R = 67.8%, G = 74.6%
- G group had greater incidence of infections and Infusion Related Reactions (IRR) than R
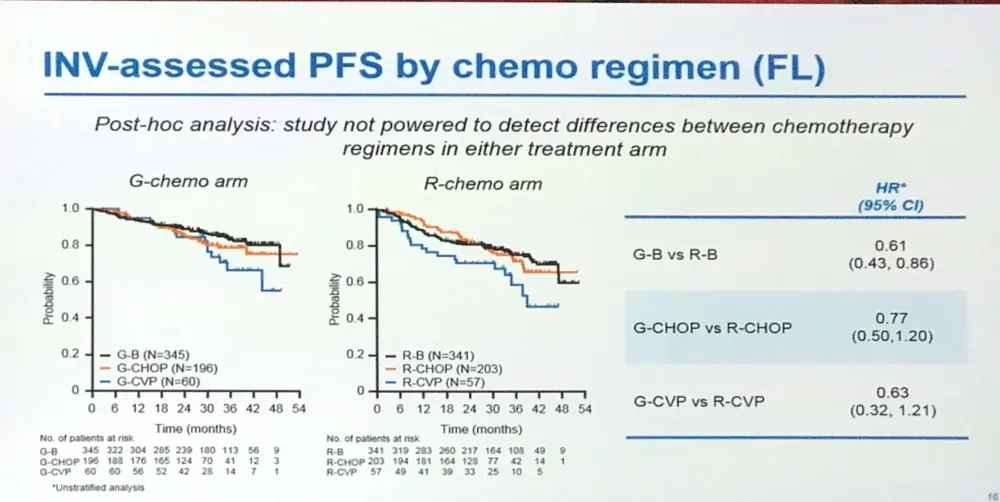
- Study was not powered to detect differences in chemotherapy regimens
Robert Marcus concluded by stating that the data showed that G-chemo with maintenance G was superior to similar R based regimen in this setting, with a “clinically meaningful improvement in PFS” (34% risk reduction) although this resulted in higher AEs. Dr Marcus recommended that G-chemo with maintenance G should now be considered as a first line treatment for patients with untreated FL.


Abstract:
Background: Immunochemotherapy induction followed by maintenance with rituximab (R) is the standard of care (SoC) for pts with advanced-stage symptomatic follicular lymphoma (FL), achieving a median PFS of 6–8 yrs and a median survival of 12–15 yrs. However, FL is incurable and most pts eventually relapse. Relapse occurs in 30% of pts within 3 yrs, and is associated with a poor prognosis. Obinutuzumab (GA101; GAZYVA/GAZYVARO; G) is a glycoengineered type II anti-CD20 monoclonal antibody with enhanced direct cell killing and antibody-dependent cellular cytotoxicity that has promising activity and manageable toxicity when combined with chemotherapy in relapsed indolent non-Hodgkin lymphoma (iNHL). We report the results of GALLIUM (NCT01332968), a global, open-label, randomized Phase 3 study comparing the efficacy and safety of R or G with chemotherapy followed by maintenance as first-line treatment in iNHL.
Methods: Pts entered were aged ≥18 yrs with previously untreated FL (grades 1–3a) or chemotherapy-naïve marginal zone lymphoma (MZL), stage III/IV disease or stage II with tumor diameter ≥7cm, ECOG PS 0–2, and requiring treatment according to GELF criteria. CHOP, CVP, or bendamustine (B) were allocated according to center (FL) or pt (MZL). Pts were randomized 1:1 and stratified by chemotherapy, FLIPI or IPI group, and geographic region to R 375mg/m2 on Day (D) 1 of each cycle or G 1000mg on D1, 8 and 15 of Cycle 1 and D1 of subsequent cycles, for either 8 x 21-day cycles (CHOP and CVP) or 6 x 28-day cycles (B). Pts with a CR or PR at the end of induction (EOI) as per modified Cheson criteria received R or G every 2 mo for 2 yrs or until disease progression. The primary endpoint was investigator (INV)-assessed PFS in FL pts. At final analysis, based on 370 PFS events having occurred, the study would have 80% power to detect a HR of 0.741. The current data are from a planned interim efficacy analysis done when 67% of the 370 PFS events had occurred (cut-off date: January 31, 2016) after which the study was unblinded on IDMC recommendation. Efficacy was assessed in all randomized FL pts. Safety was assessed in all FL pts who received any study treatment.
Results: Results are reported for 1202 FL pts (R-chemo, 601; G-chemo, 601) with a median age of 59 yrs (53.2% female); data for 195 MZL pts will be reported elsewhere. Treatment arms were well balanced by disease stage (Ann Arbor: I, 1.5%; II, 7.1%; III, 34.9%; IV, 56.5%) and prognostic factors (FLIPI: 0+1, 21.0%; 2, 37.2%; ≥3, 41.8%; FLIPI-2: 0, 9.1%; 1+2, 50.3%; ≥3, 40.6%). Chemotherapy received was B in 57.1% of pts, CHOP in 33.1%, and CVP in 9.8%. CR and ORR at EOI based on CT/NMR imaging were similar for the two arms (Table 1). After a median follow-up of 34.5 mo (range, 0–54.5), there was a 34% reduction in the risk of progression or death (HR, 0.66; 95% CI, 0.51, 0.85; p=0.001; Figure; Table 1). Although medians have not been reached in GALLIUM or PRIMA, the observed HR of 0.66 would translate to a 1.5x longer median PFS for G-chemo than R-chemo, and to an estimated 3 yr improvement in the G arm if a median PFS of 6 yrs was assumed in the R arm. Three-yr INV-assessed PFS rates were: G-chemo, 80.0% (95% CI, 75.9%, 83.6%); R-chemo, 73.3% (95% CI, 68.8%, 77.2%). A consistent benefit in favor of G-chemo was also seen for PFS assessed by Independent Review Committee (IRC), as well as other time-to-event endpoints (Table 1). Subgroup analyses were broadly consistent with the primary analysis. At the time of the analysis, 35 pts (G-chemo; 5.5%) and 46 pts (R-chemo; 8.7%) had died (HR for overall survival, 0.75; 95% CI, 0.49, 1.17; p=0.210; Table 1). G-chemo pts had a higher frequency of grade 3–5 AEs (74.6%) and SAEs (46.1%) than R-chemo pts (67.8% and 39.9%, respectively). The frequency of fatal AEs was similar (G-chemo, 4.0%; R-chemo, 3.4%). AEs led to treatment discontinuation in 16.3% pts (G-chemo) and 14.2% pts (R-chemo) in the absence of disease progression. Safety results are summarized in Table 2. The median decrease in IgG levels at the EOI treatment was similar in the two arms (Table 2).
Conclusion: In pts with previously-untreated FL, G-based immunochemotherapy and maintenance resulted in a clinically meaningful improvement in PFS, with a 34% reduction in the risk of a PFS event relative to R-based therapy. Frequency of some AEs, e.g. infusion-related reactions (IRRs), cytopenias, and infections, was higher with G. These data support G-chemo becoming a new SoC in previously untreated pts with FL.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

