All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
ESMO 2016 | Educational Session: CLL – Moving towards chemotherapy free approaches
At this year’s ESMO conference, during an educational session, Professor Paolo Ghia of the Vita-Salute San Raffaele University in Milan gave a presentation entitled “CLL: Moving towards chemotherapy-free approaches.”
Professor Ghia started the presentation by commenting on the marked evolution of treatments for Chronic Lymphocytic Leukemia (CLL) over the last five decades; from single alkylating agents to more complex chemoimmunotherapies using bendamustine and rituximab (BR) and fludarabine/cyclophosphamide/rituximab (FCR) regimens.
Given the increased range of treatment choices in CLL, finding the best treatment option can be challenging. Ghia then presented the ESMO 2015 treatment guidelines for first line CLL by B. Eichhorst et al. on the parameters that should be considered for each treatment scenario, these include:
- Whether it is early stage with or without active disease
- The type of mutation 17del or TP53
- The fitness of the patient

Note: In September 2016, ESMO published an eUpdate on their CLL recommendations.
Professor Ghia then discussed data which he had published in 2012 at ASH on the role of Minimal Residual Disease (MRD) in guiding therapy and predicting prognosis. According to Ghia, over the last 20 years the efficacy of therapies for CLL has increased dramatically, leading to progressively higher percentages of clinical Complete Remissions (CR). As such, this has led to debates regarding the importance of MRD. In this publication, Ghia affirms that MRD is a strong predictor for clinical outcome and states that emerging data indicate that MRD status during and at the end of treatment is one of the most powerful predictors of Progression Free and Overall Survival (PFS; OS). Moreover, data from Thompson et al. suggests that achieving bone marrow MRD-negative CR is associated with superior PFS and OS; MRD status is the single best post-treatment predictor of long-term outcomes after chemoimmunotherapy.
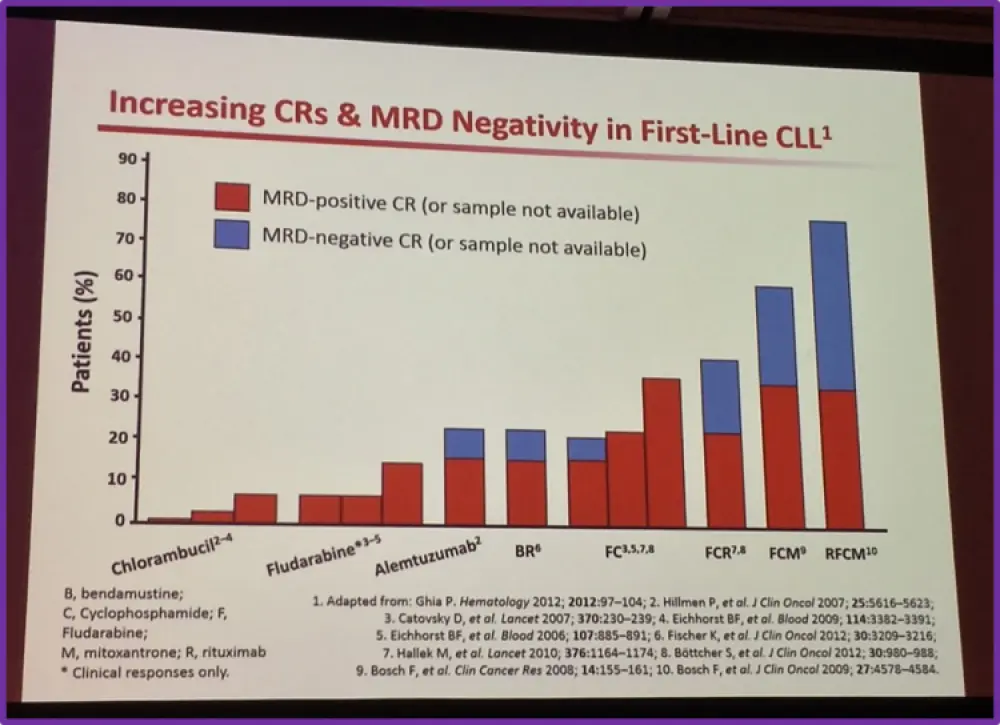
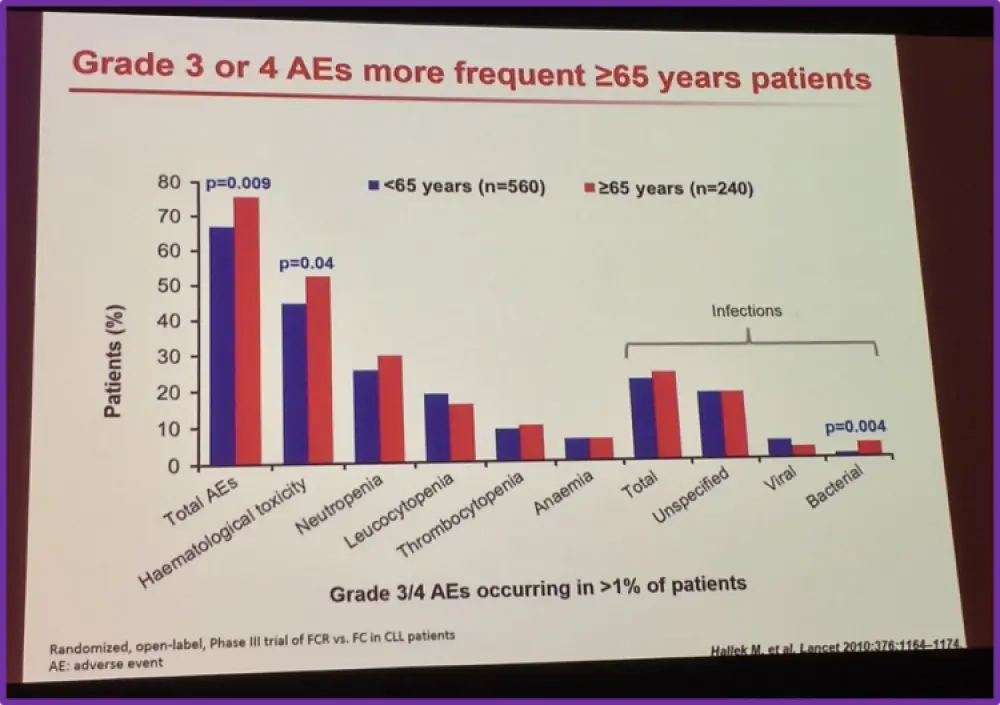
Ghia then went on to explain the rationale for moving away from chemoimmunotherapy despite the reported efficacy rates. According to Brown et al., the side effects of chemoimmunotherapy are serious, ranging from myelosuppression to the development of other myeloid malignancies such as Acute Myeloid Leukemia (AML). There have been reports of 2.39 times increased episodes of secondary cancers following treatment with FCR regimens. In particular, Adverse Events (AEs) are at a higher frequency in elderly patients. Elderly patients still remain a challenging patient group to treat. In particular, elderly patients are prone to more grade 3–4 AEs (Hallek et al. Lancet. 2010).
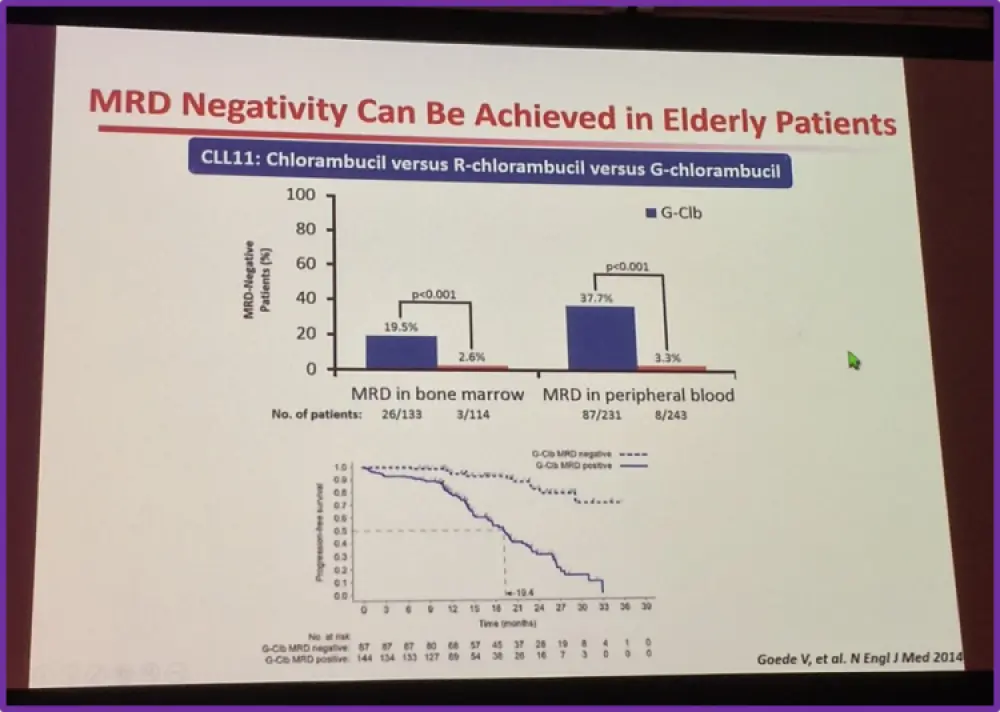
However, Ghia presented data from Goede et al. who published a phase 3, randomized trial in the New England Journal of Medicine, which analyzed whether anti-CD20 antibody-based chemoimmunotherapy (with chlorambucil as the chemotherapy backbone) would be beneficial in patients with CLL and co-existing conditions and whether targeting of the CD20 antigen by obinutuzumab could improve outcomes as compared with rituximab. Goede et al. reported that obinutuzumab-chlorambucil, as compared with rituximab-chlorambucil, resulted in higher rates of overall, complete, and molecular responses. Professor Ghia concluded from this data that MRD-negative status could be achieved in elderly patients.
The role of emerging novel therapeutic agents was then explored. More recently, there has been increased development in targeted therapy, contrary to chemotherapy which work by attacking rapidly growing cells in general (including cancer cells), these agents attack one or more specific targets on or in cancer cells. These novel molecules in CLL include the following: Bruton’s Tyrosine Kinase (BTK) inhibitors, PI3K inhibitors and BCL-2 inhibitors:
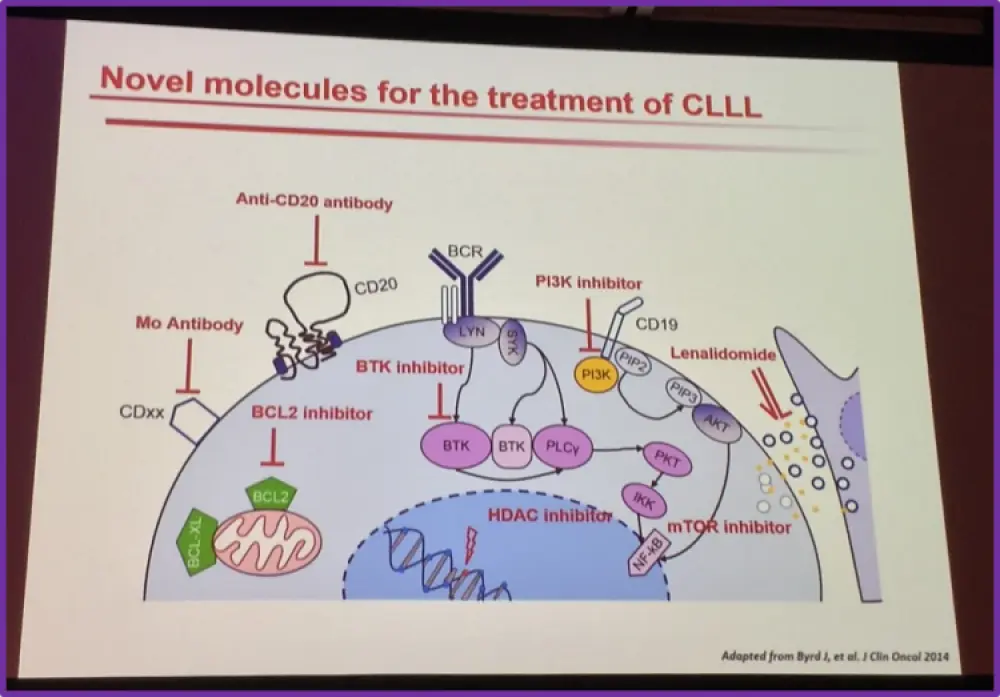
- Ibrutinib (Imbruvica)
- Idelalisib (Zydelig)
- ABT-199 (venetoclax)
Ibrutinib was the first-in-class BTK inhibitor. Ghia shared data on the efficacy of ibrutinib versus chemoimmunotherapy. Burger et al. in 2015 published an international, open-label, randomized phase 3 trial to compare two oral agents, ibrutinib and chlorambucil, in previously untreated older patients with CLL or small lymphocytic lymphoma (RESONATE-2 ClinicalTrials.gov number, NCT01722487). Ghia highlighted the differences in Overall Response Rate (ORR) for patients treated with ibrutinib versus chlorambucil at 8 months. However, Burger et al. reported that the estimated ORR at 24 months was 98% with ibrutinib versus 85% with chlorambucil. The results of this study led to ibrutinib being granted FDA approval for use in patients with previously untreated CLL.
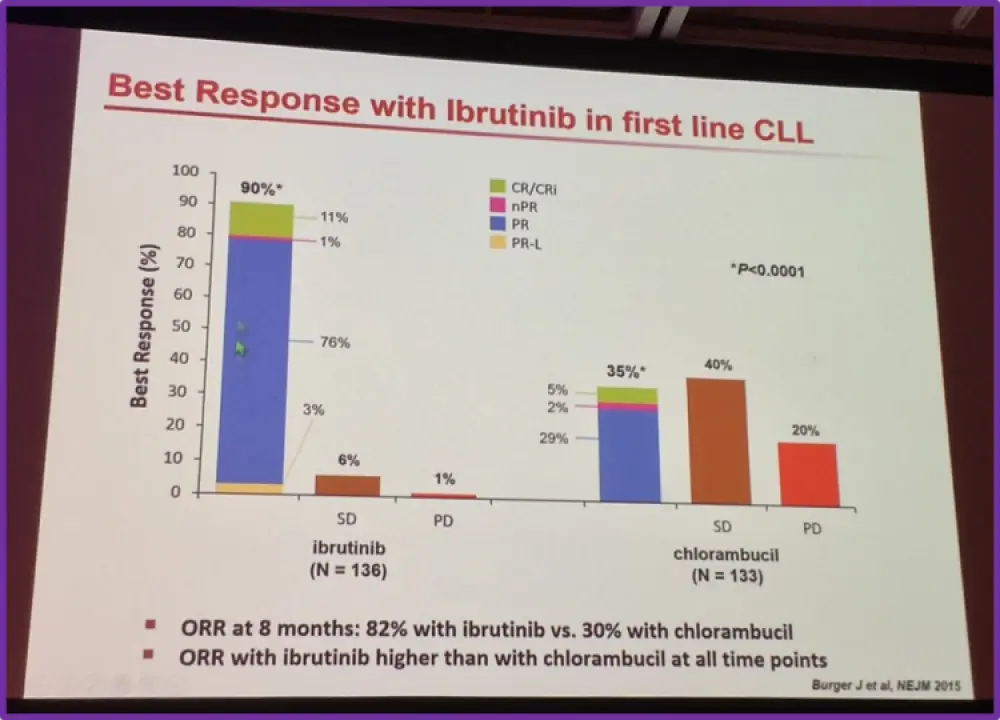
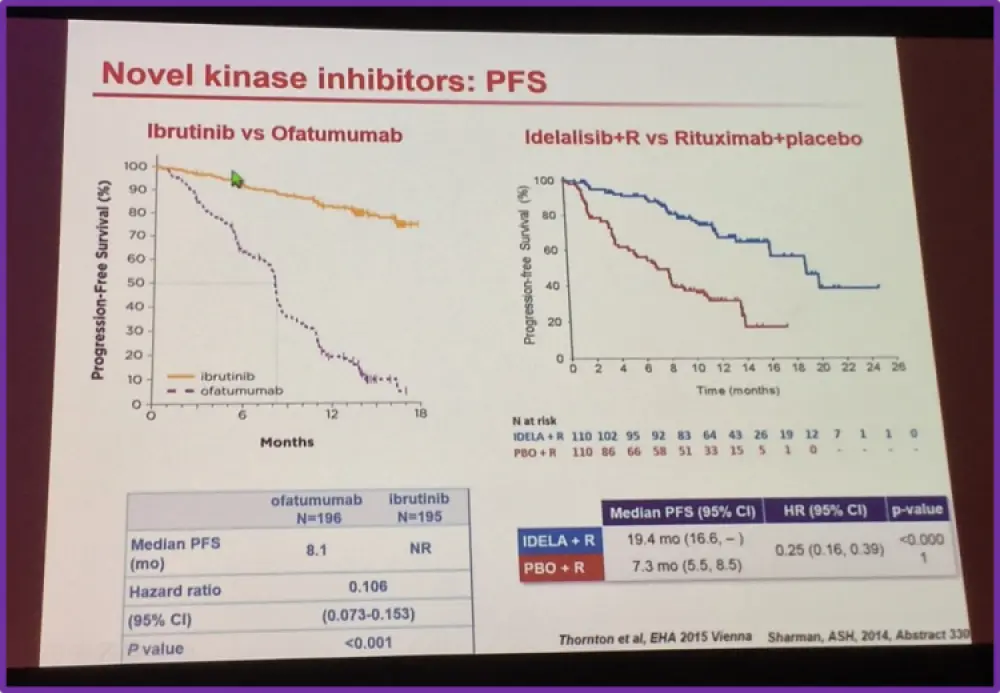
Ghia also presented efficacy data of ibrutinib compared to other chemoimmunotherapy in terms of PFS.
Idelalisib is a PI3K inhibitor that has been approved by the FDA for use in CLL. Brown et al. published findings of their phase 1 trial of idelalisib in patients with relapsed CLL in Blood in 2014. Those treated with idelalisib had a 72% ORR and the median PFS for all patient cohorts in the study was 15.8 months.
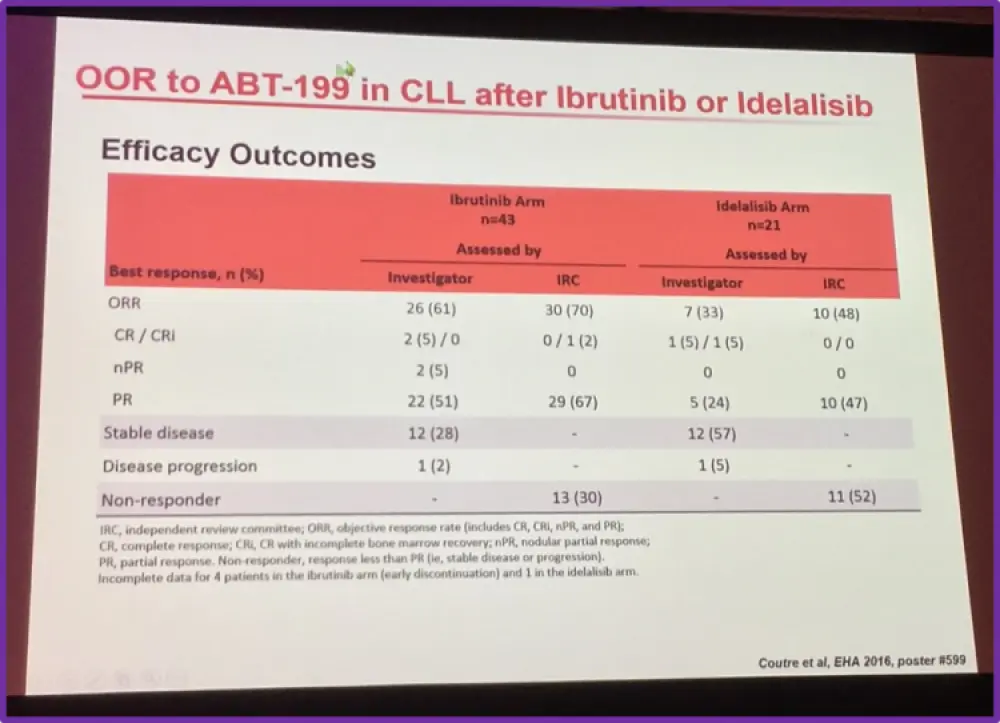
Then, attention was turned to another novel agent, venetoclax (ABT199). The BCL-2 inhibitor is expected to receive its FDA approval this year. The data to date has demonstrated increased efficacy and fewer episodes of thrombocytopenia. Data on the efficacy of ABT199 in comparison to ibrutinib and idelalisib were presented (Coutre et al. EHA 2016; poster 599).
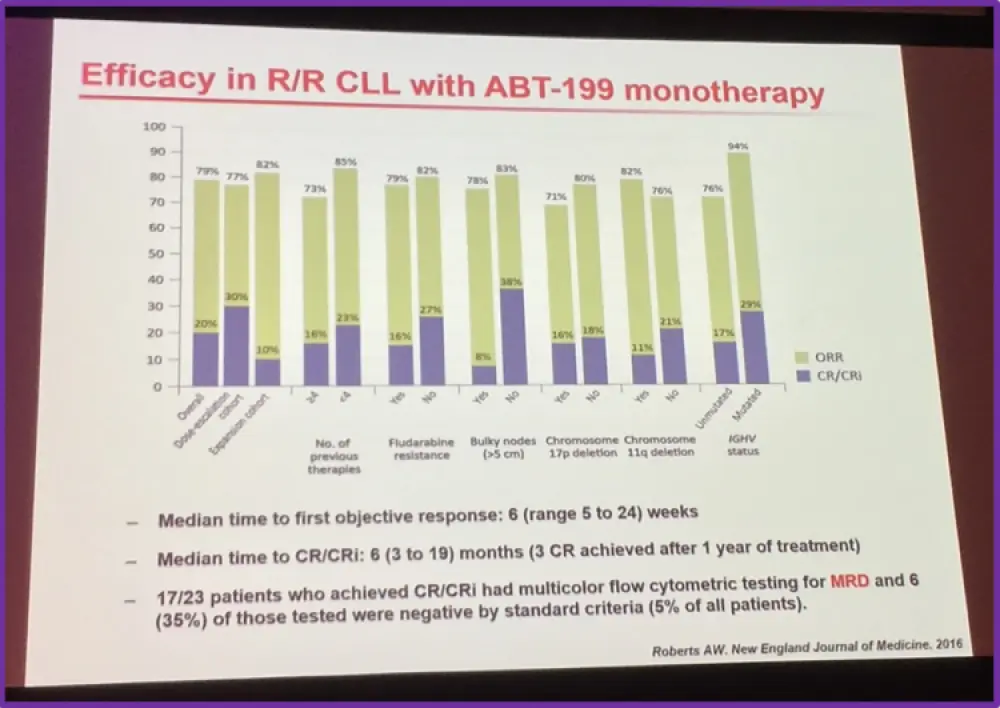
Professor Ghia also presented data by Roberts et al. who published in the New England Journal of Medicine, 2016, the results of a phase 2 trial on the use of ABT-199 as monotherapy in R/R CLL. They reported a PFS at 12 months of 72% and an estimated OS of 87%.
Then Professor Ghia presented data from a ground-breaking study of rituximab in combination with ABT-199.This data taken from a phase 1b, open-label, dose-escalation, multicenter, international study which assessed the maximum tolerated dose and safety of venetoclax combined with rituximab in patients with Relapsed/Refractory (R/R) CLL or Small Lymphocytic Leukemia (SLL). An abstract on this was presented at EHA 2015 (abstract number S431). Of the 49 patients evaluated, the ORR was 86% and CR was 47%. In addition, MRD-negative status in the bone marrow was reported in 13 of the 20 (65%) patients who achieved CR/CRi and in 24 out of 49 (49%) patients overall.
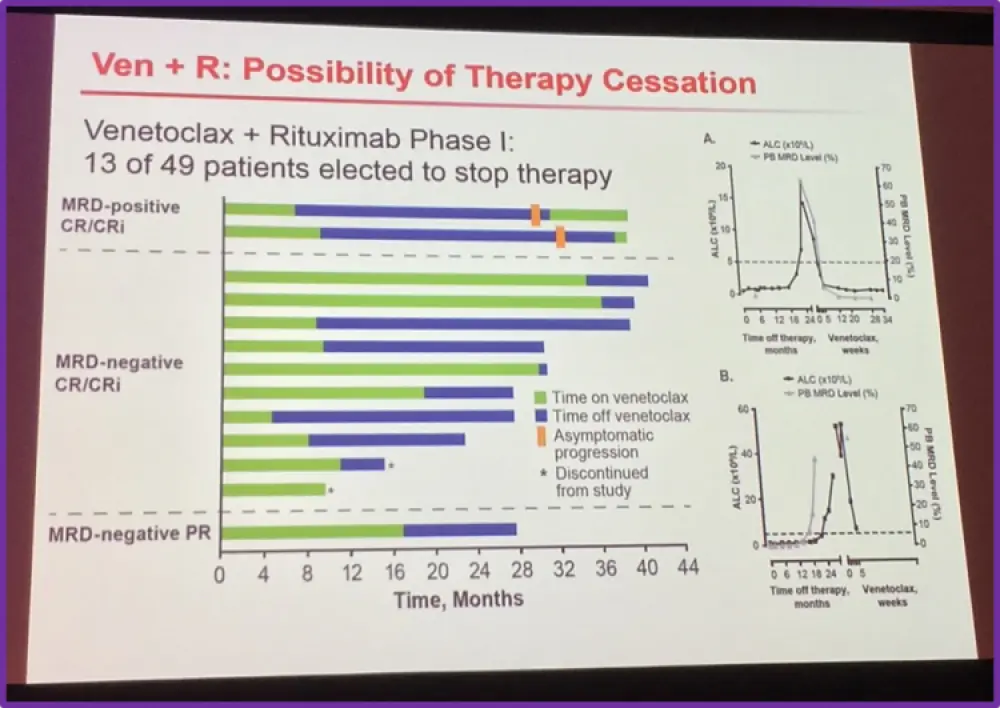
These data provide a starting point on the discussion of treatment cessation in CLL with novel targeted therapy. Although, a recent publication from Brown et al. states that more information is still required before this can become a routine practice and they pose the following questions:
- Is there a clinical marker such as the absence of MRD that would suggest appropriate discontinuation?
- How long should patients remain on a kinase or BCL-2 inhibitor?
- For patients with low risk disease receiving single therapy, should you stop treatment and observe at some point?
This educational session was concluded on the thought of whether these advancements have led to the end of chemotherapy? We await the results of a planned phase 3 multicenter, randomized, prospective, open-label trial of standard chemoimmunotherapy (FCR/BR) versusrituximab plus venetoclax (RVe) versus obinutuzumab (GA101) plus venetoclax (GVe) versus obinutuzumab plus ibrutinib plus venetoclax (GIVe) in fit patients with previously untreated CLL without del (17p) or TP53 mutation.

Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average vein-to-vein time when treating patients with DLBCL with a reimbursed CAR T-cell therapy (from apheresis to infusion)?

