All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
ESMO 2016 | Educational Session – Waldenström Macroglobulinemia Overview
Professor Meletios A. Dimopoulos from the National and Kapodistrian University of Athens, Greece, presented an educational session on Waldenström Macroglobulinemia (WM) during the ESMO congress in October 2016 in Copenhagen, Denmark.
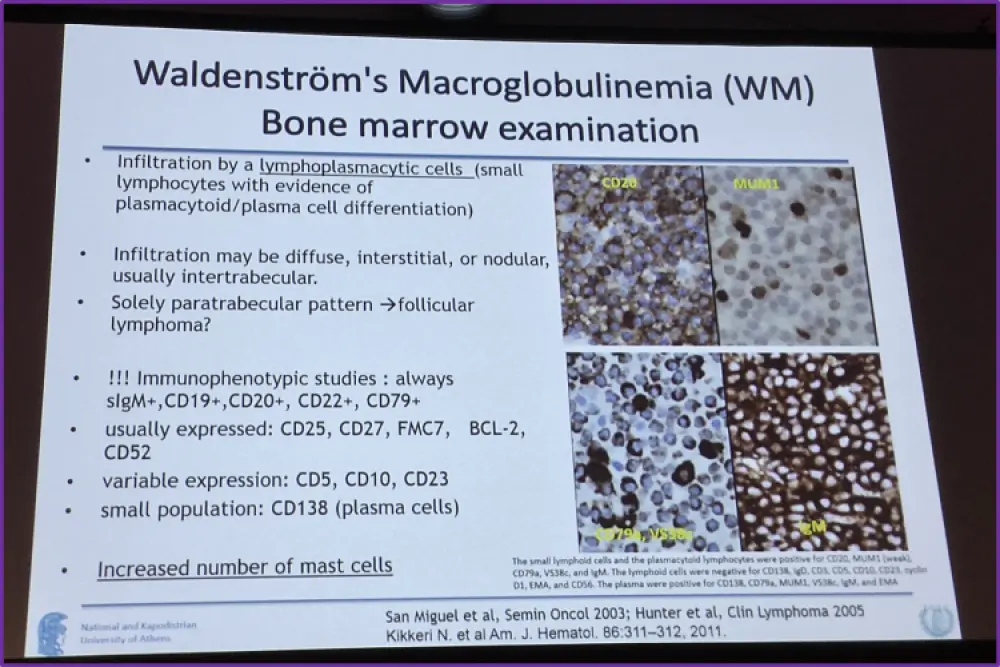
The session began with explaining the background and clinical origins of the classification of WM, before moving on to discuss the diagnostic criteria used currently. These criteria were: any concentration of monoclonal IgM, lymphoplasmacytic cells found within bone marrow, immunophenotypes of surface IgM+, FMC7+ CD103-, CD138-, CD5+, CD10-, CD19+, CD20+, CD22+, CD23-, CD25+, and CD27+. The last two of these criteria are considered supportive but not necessary for diagnosis.
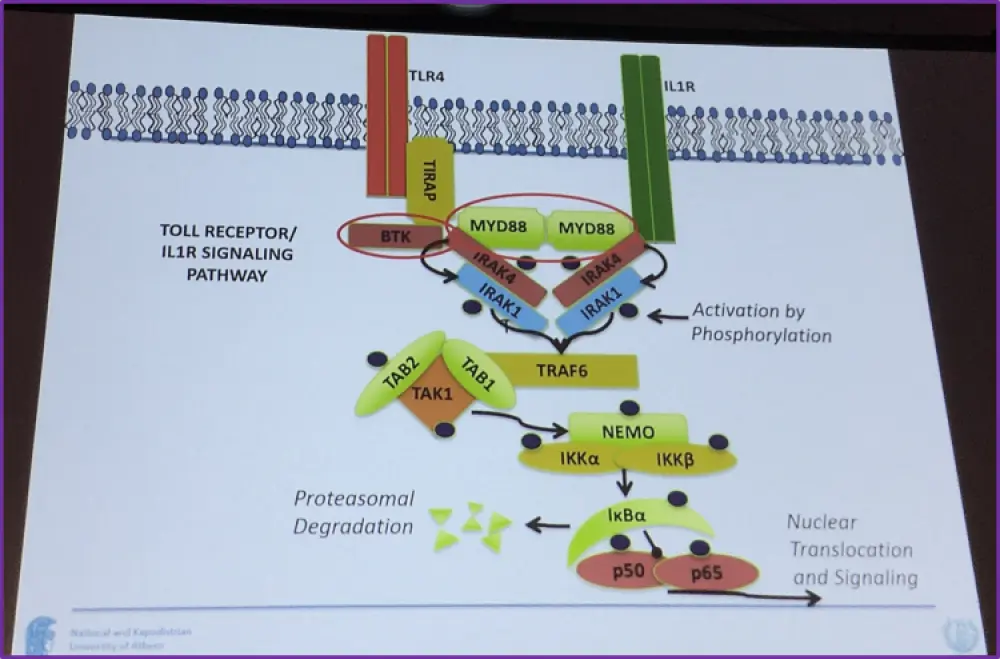
The genetics of WM were the next topic of discussion and data presented stated that 91% of patients with WM have the MYD88 somatic mutation L265P (Treon, et al. 2012). MY88 protein is involved in the phosphorylation of IRAK4, interacts with IL1R, its downstream of TLR4, and upstream of NEMO, p50 and p65. Additionally, a somatic C-tail WHIM-like mutation in CXCR4 is present in 27–31% of WM patients.
Data presented from Treon et al. at ICML 2011 showed that of over 1,000 patients diagnosed with WM, 26.1% had a first or second degree relative with a form of B-cell lymphoproliferative disorder.
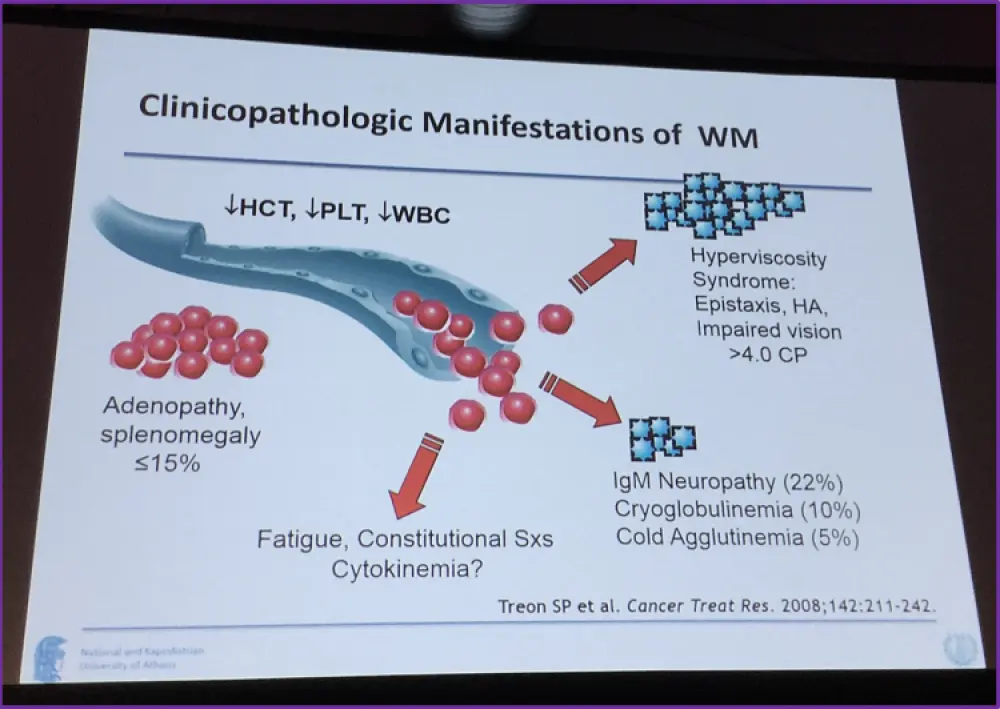
The next topic covered was the clinicopathologic manifestations of WM, shown below, along with the recommendations for the initiation of therapy for WM, noting that IgM levels by itself is not sufficient for the initiation of therapy.

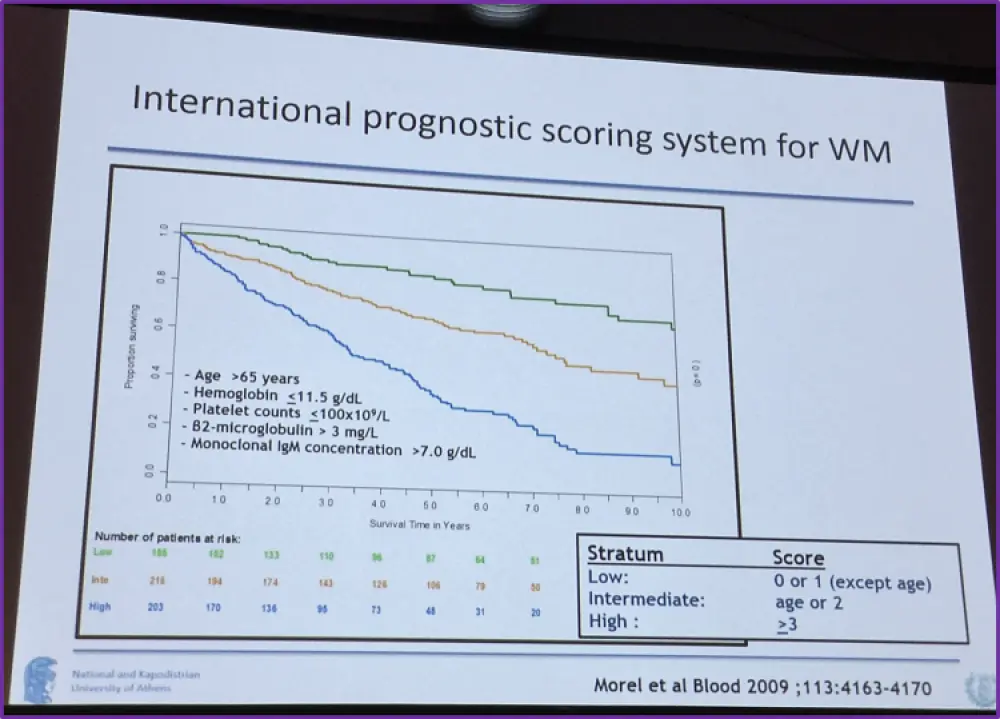
Morel et al. in 2009 presented an international prognostic scoring system for WM in which they found five factors affecting prognosis and these were: age >65 years, monoclonal IgM > 7.0 g/dL, platelet count ≤ 100 x 109/L, β2-microglobulin > 3 mg/L, and hemoglobin ≤11.5 g/d. Low-risk patients presented with no or one of these factors, intermediate risk with two factors or only age >65 years, and high-risk patients were classified as having more than two of these factors. The survival rates are shown in the graph below.
A summary slide of the challenges facing the management of WM was presented. Outlined were the facts that WM is a rare disease, mostly in the elderly, and that eradication of the disease is not currently realistic. Furthermore, there are ‘hardly any’ drugs approved for treating WM, large trials are difficult and that for most patients, an intense treatment is not feasible.
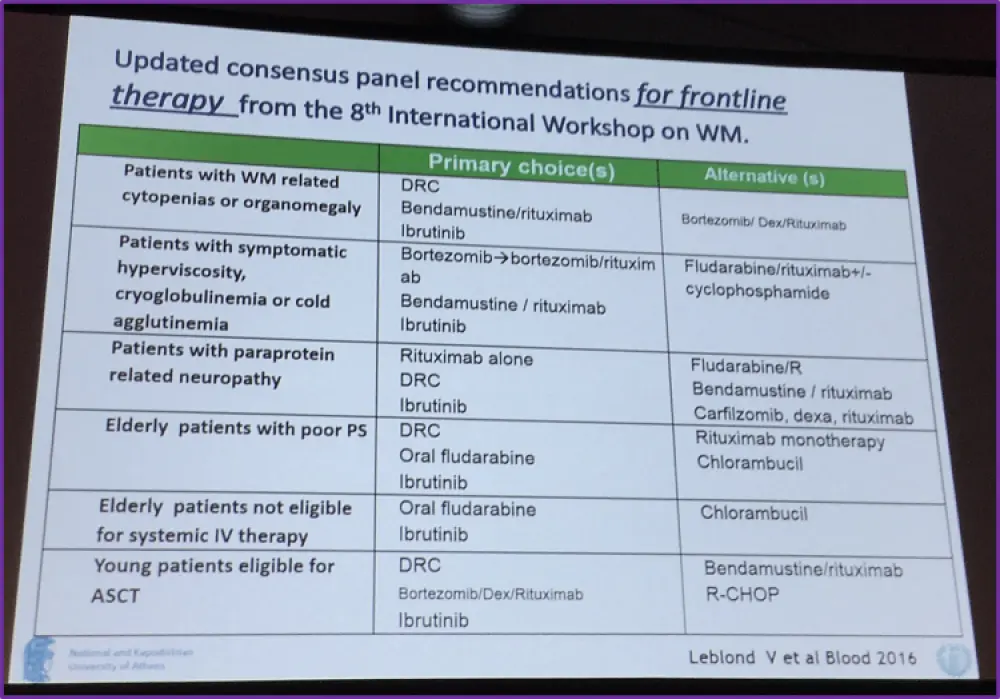
The recommended frontline therapy options, from the 8th International Workshop for WM, were then summarized on the two slides pictured below:

Finally, Professor Dimopoulos summarized the talk with the following points:
- Diagnostic role of MYD88 L265P: is there MYD88 WT WM?
- Individualized therapy based on clinical demands: is it feasible?
- Short duration (~4 months) of rituximab-based regimens for most patients with untreated WM (DRC, BR, BDR in selected patients) --> high ORR, PFS >3 years
or
- BTK inhibitor oral therapy for prolonged periods (how long?)
- Can we choose therapy based on mutational status of MYD88 and CXCR4?
- Ibrutinib may be a primary choice for selected patients (i.e. high IgM, cytopenias, neuropathy) - may not be a good choice if bleeding diathesis
- For patients with rituximab-refractory disease, ibrutinib is probably the best option
- Limited data exists for patients who fail ibrutinib, but proteasome inhibitors (bortezomib) may be active
- Ibrutinib therapy is long lasting and expensive - IgM increases may occur if discontinued
Data for the efficacy of the different treatment options discussed for WM in this talk are presented in the following publications: Dimopoulos, et al. Blood. 2014, Kastritis, et al. Blood. 2015, Rummel, et al. Lancet. 2013, Dimopoulos, et al. Blood. 2013, Treon, et al. Blood. 2014, and Treon, et al. NEJM. 2015.
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average vein-to-vein time when treating patients with DLBCL with a reimbursed CAR T-cell therapy (from apheresis to infusion)?

