All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
ICML 2017 | Chemotherapy-free combinations for patients with B-cell NHL – Focus On Session
One of the “Focus On” Sessions at the 14th International Conference on Malignant Lymphoma (ICML) in Lugano, Switzerland, was based around chemotherapy-free combinations in patients with B-cell NHL.
The session was chaired by Sonali M. Smith from the University of Chicago, Chicago, IL, USA.
Abstract 035
The first talk was scheduled to be given by Peter Martin, MD; however, due to travel issues, the talk was instead given by John P. Leonard, MD, both from Weill Cornell Medical College. The presentation conveyed final results from the Alliance/CALGB 50803 phase II study of lenalidomide plus rituximab for newly diagnosed Follicular Lymphoma (FL) patients (NCT01145495).
Previously, it has been reported by SAKK that 8 doses of rituximab achieved a median EFS of 36 months in responders. Based on this, CALGB conducted two trials of rituximab in combination in patients with newly diagnosed FL: the 50402 study of rituximab plus galiximab (3-year PFS 48%; 75% in FL-IPI 0–1) and the 50701 study of rituximab plus epratuzumab (3-year PFS 60%; 85% in FL-IPI 0–1). Another CALGB study has evaluated lenalidomide and rituximab in previously treated FL patients and found a median EFS of 2 years.
The rationale for the CALGB 50803 trial was that the rituximab-lenalidomide combination had been evaluated in a single center study and so P. Martin and colleagues wanted to evaluate it in a multicenter study. Based on toxicity from the previous single-center study, the dose was reduced to 20mg/day on days 1–21 of a 28-day cycle for 12 cycles. Rituximab administered 4-times weekly on cycle 1 and day 1 of cycle 4, 6, 8, and 10. Patients with untreated FL, grade 1–3a, stage 3–4 or bulky stage 2, and FL-IPI 0–2 were eligible. The primary outcome measure was Complete Response (CR) and polymorphisms in FcgR2A and FcgR3A were evaluated.
Overall, 66 patients were enrolled with a median age of 53 years (range, 32–79); of these, 65 were included in the final analysis (one patient never started treatment).
|
Characteristics |
N = 66 |
|---|---|
|
Age |
53 (32–79) |
|
Sex M vs. F |
32 vs. 34 |
|
FL-IPI 0–1 vs. 2 vs. 3 |
21 vs. 43 vs. 2 |
|
Grade 1 v. 2 vs. 3a |
39 vs. 21 vs. 4 |
|
Non-bulky vs. bulky |
50 vs. 15 |
|
FCGR3A 158F vs. 158F/V vs. 158 V |
22 vs. 27 vs. 10 |
|
FCGR2A 131H vs. 131R/H vs. 131R |
18 vs. 28 vs. 13 |
In total, 51 patients (78.5%) completed 12 cycles of lenalidomide. Reasons for early termination included AE (n = 6), patient refusal (n = 6), and disease progression (n = 2). Grade 3–4 neutropenia was reported in 21% of patients. Grade 3 infections were observed in 20% of patients, and grade 3 rash, fatigue, hyperglycemia, hypophosphatemia, and hypertension was reported in 9%, 6%, 6%, 6%, and 6% of patients, respectively. Grade 4 decrease in platelets was seen in 2% of patients. Notable AEs included thromboembolism (grade 1 n=1; grade 2 n=2), febrile neutropenia (grade 3 n=1), and Tumor Lysis Syndrome (grade 3 n=2). No grade 4 non-hematologic AEs, secondary malignancies, or tumor flare reported.
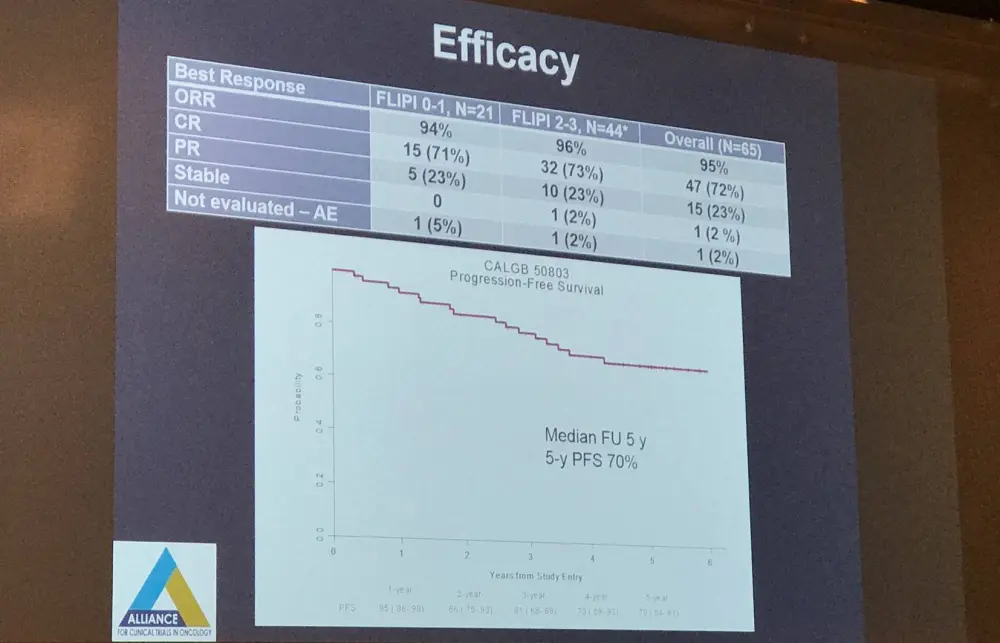
Mean Circulating Endothelial Cells (CECs) and Hematopoietic Progenitor Cells (HPCs) substantially decreased versus baseline (P < 0.01 in both), while Endothelial Progenitor Cells (EPCs) remained stable (P = 0.88). No association was found between decrease in CEC/HPC or FcgR2A/FcgR3A polymorphism and CR. Progression has taken place in 16 patients, 7 of which had a best response of CR, 8/9 patients with a best response of PR, and one patient with SD. After a median follow-up of 5 years, the 2-, 3-, 4-, and 5-year PFS were 86%, 81%, 73%, and 70%. Furthermore, no association was found between FL-IPI and CR rate or PFS. Overall survival is 100%.
In conclusion, this multicenter, phase II trial found that rituximab-lenalidomide is highly active as a first-line treatment options in patients with low- and intermediate-risk FL-IPI. Moreover, the regimen displayed a tolerable safety profile. Lastly, no association was found between FcR polymorphism/change in angiogenic markers and CR/PFS.
Abstract 036
Gilles Salles, MD, PhD, South Lyon Hospital Complex, Lyon, France, gave the second talk during this session, focused on results from the phase II, single-arm L-MIND study (NCT02399085) assessing MOR208 combined with lenalidomide in Relapsed or Refractory (R/R) Diffuse Large B-Cell Lymphoma (DLBCL) patients. We interviewed him while at the meeting; watch it here.
Abstract 037
The next presentation was given by Franck Morschhauser from Centre Hospitalier Régional Universitaire de Lille, Lille, France, and contained results from a phase I/II trial by LYSA investigating obinutuzumab (GA) plus lenalidomide (LEN) for R/R FL (NCT01582776).
LEN has shown synergy with rituximab (R2) in patients with R/R NHL (Leonard et al. JCO. 2015) and in newly diagnosed FL patients (Fowler et al. Lancet Oncol. 2014). GA is a glycoengineered anti-CD20 monoclonal antibody which has demonstrated increased Antibody Dependent Cellular Cytotoxicity and direct cell death induction than rituximab; therefore, GALEN may potentially be more efficient than R2.
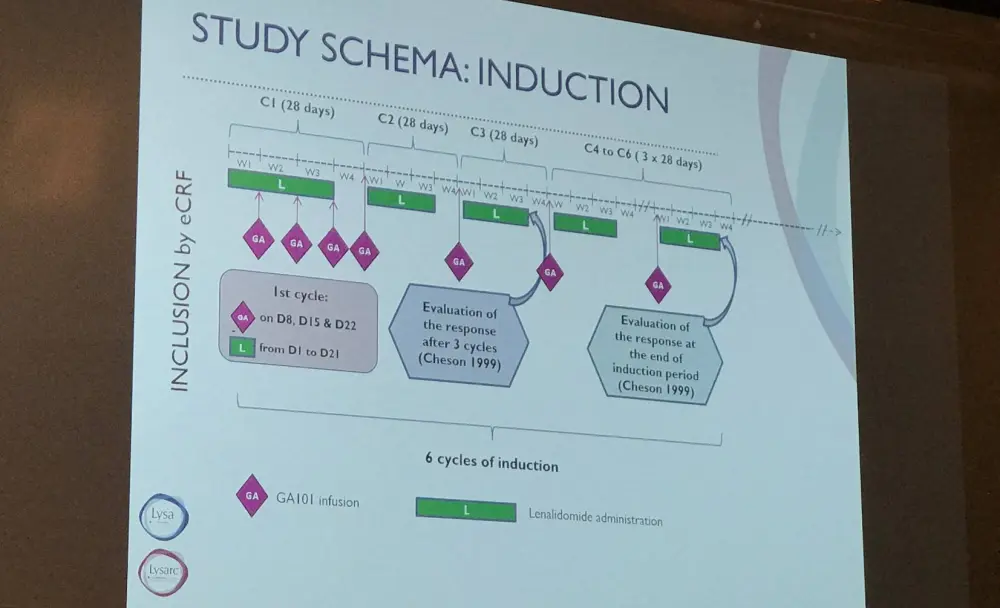
Patients were included if they had R/R FL, stage 1–3a disease, received one or more rituximab-containing regimens, and an ECOG PS ≤2. The primary outcome measure of the study was ORR at the end of induction, and secondary endpoints were efficacy (CR, PFS, OS) and safety (AEs, Secondary Primary Malignancies [SPMs]).
Induction therapy consisted of LEN 20mg (92%) or 10mg (8%) on day 1–21 of a 28-day cycle for the first cycle and on day 2–22 of a 28-day cycle from cycles 2 to 6. GA was administered at a dose of 1000mg by IV on days 8, 15, and 22 of cycle 1 and then day 1 of cycles 2 to 6. Patients who responded were then administered maintenance therapy of 12 cycles of LEN at 10mg on day 2–22 every 28 days and GA 1000mg every 8 weeks for 12 cycles until progression or unacceptable toxicity.
Between June 2014 and December 2015, 89 patients were enrolled with a median age of 64 years and a median of 2 previous lines of therapy (range, 1–7); 26.7% were refractory to a rituximab-containing regimen or last previous therapy. Of those enrolled, 86 were assessable for efficacy and 88 were assessable for safety. After a median follow-up of 18.1 months, 75 patients (87.2%) had completed induction and 67 (78%) went on maintenance (ongoing in 45 patients).
The most frequent AEs (>20% of patients) during induction (all Gr / Gr 3–4) were gastrointestinal disorders (76.1% / 2.3%), infections (62.5% / 6.8%), asthenia (52.3% / 2.3%), neutropenia (30.7% / 28.4%), muscle spasms (30.7% / 0%), and cough (20.7% / 0%). Febrile neutropenia was reported in 3.4% of patients. Other AEs of special interest were infusion related reactions (13 patients; 14.8%), tumor flare (1 patient; 1.1%), venous thrombosis (1 patient; 1.1%), and SPMs (3 patients; 5 Basal Carcinomas and 1 Myelodysplastic Syndrome).
Response rates and outcomes found for the GALEN regimen are listed in the table below:
|
|
|
All patients (n=86) |
Early relapse patients (within 2 years of first-line treatment; n=24) |
Refractory patients (n=23) |
|---|---|---|---|---|
|
IWG 1999 |
ORR, % (95% CI) |
80.2 (70.2–88.0) |
70.8 (48.9–87.4) |
60.9 (38.5–80.3) |
|
CR/CRu, % (95% CI) |
39.5 (29.1–50.6) |
33.3 (15.6–55.3) |
34.8 (16.4–57.3) |
|
|
IWG 2007 |
ORR, % (95% CI) |
74.4 (63.8–83.2) |
66.7 (44.7–84.4) |
56.5 (34.5–76.8) |
|
CR, % (95% CI) |
44.2 (33.5–55.3) |
54.2 (32.8–74.4) |
30.4 (13.2–52.9) |
|
|
1-year PFS |
% (95% CI) |
75.5 (64.2–83.7) |
74.8 (52.2–87.8) |
65.2 (42.3–80.8) |
|
1-year OS |
% (95% CI) |
88.8 (79.5–94.0) |
86.9 (64.6–95.6) |
71.5 (47.1–86.1) |
Franck Morschhauser concluded this abstract presentation by stating that GALEN was highly effective in patients with R/R FL. It appeared superior to R2, particularly in patients with POD24 based on historical comparison to the MAGNIFY study. Moreover, the regimen appears safe and did not demonstrate any unexpected toxicities. Morschhauser emphasized that longer follow-up is required to assess the full impact of the maintenance therapy in those patients who responded to induction.
Abstract 038
Jean-Marie Michot from the Institut Gustave Roussy, Villejuif, France, gave the next talk titled which included results from a phase IB study assessing the combination of CC-122 and obinutuzumab in R/R patients with NHL (NCT02417285).
CC-122 is a cereblon modulating agent and has immunomodulatory effects on T- and NK-cell function. The combination of CC-122 and obinutuzumab has shown synergism pre-clinically in FL and additive anti-lymphoma effects in DLBCL compared to either as a single agent.
Primary endpoints of this phase IB trial included safety/tolerability, Non-Tolerated Dose (NTD), and Maximum Tolerated Dose (MTD) of CC-122 when co-administered with obinutuzumab. Secondary endpoints included ORR, DoR, PFS, and CC-122 PK. Response was assessed per Cheson 2007 criteria every 2 cycles to cycle 6, every 3 cycles to cycle 12, and every 6 cycles thereafter.
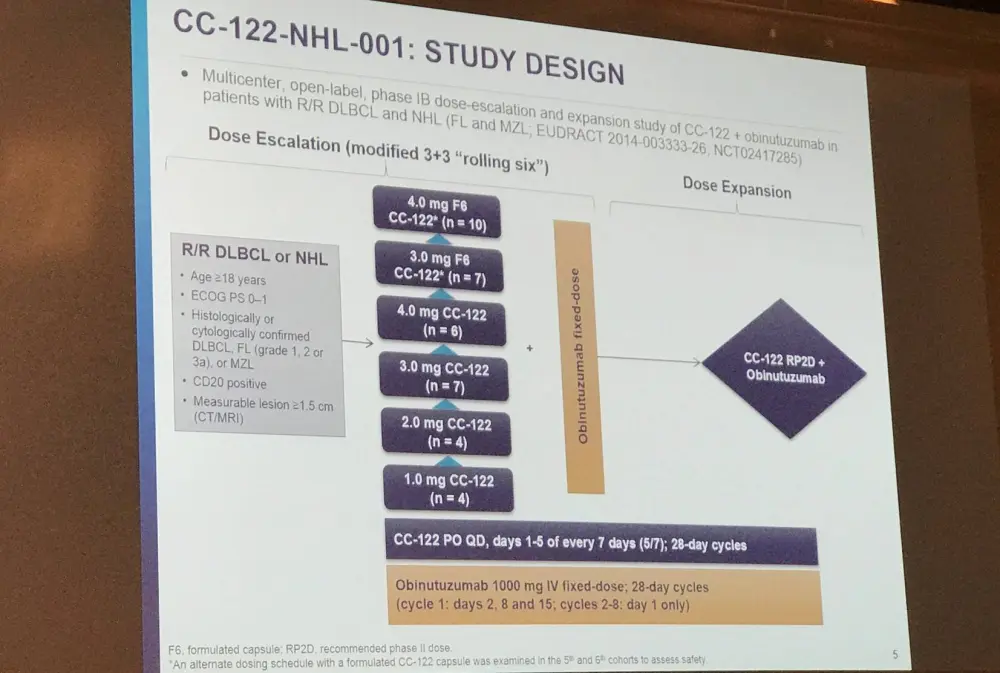
Overall, 34 patients with R/R B-cell NHL (18 DLBCL [8 transformed FL], 15 FL, 1 MZL) were enrolled. At study entry, median age of patients was 60 years (range, 26–81), 68% were male, and 76% had stage III/IV disease. Of the 16 FL/MZL patients, 44% relapsed less than one year following first-line therapy. The median number of prior regimens was 4 (range, 1–11) and 13 (38%) patients had received prior stem cell transplant.
The most common grade 3–4 AEs (>10% of patients) were hematological AEs: 2 patients (5%) experienced grade 4 febrile neutropenia and 2 patients have discontinued treatment due to AEs. Moreover, 2 patients experienced a Dose Limiting Toxicity (DLT): one a grade 4 neutropenia (3.0mg CC-122 plus obinutuzumab) and one grade 5 tumor flare reaction (4.0mg F4 CC-122 plus obinutuzumab).
Overall, ORR was 67% including 12 patients (32%) who achieved CR. The median time to best response was 57 days (95% CI, 56–114). Of patients receiving CC-122 doses ≥3.0mg plus obinutuzumab, 23/30 (77%, including 40% CR) responded. To date, patients receiving CC-122 at a dose of 3.0mg and higher have shown the best and more durable responses to CC-122 and obinutuzumab.
|
Outcomes by histology, n (%) |
All patients (n=38) |
DLBCL (n=19) |
FL/MZL (n=19) |
|---|---|---|---|
|
ORR |
25 (66) |
9 (47) |
16 (84) |
|
CR |
12 (32) |
3 (16) |
9 (47) |
|
PR |
13 (34) |
6 (32) |
7 (37) |
|
SD |
4 (11) |
3 (16) |
1 (5) |
|
PD |
6 (16) |
4 (21) |
2 (11) |
|
Not evaluable/missing |
3 (8) |
3 (16 |
0 |
This abstract presentation was concluded by stating that CC-122 combined with obinutuzumab showed clinically meaningful activity in heavily treated patients with R/R DLBCL and FL/MZL. The combination was also well tolerated. CC-122 doses at ≥3.0mg plus obinutuzumab showed increased ORR and longer DoR in DLBCL and FL. The dose escalation portion of the study is complete, and the trial is now enrolling for the dose expansion phase with a recommended phase II dose of 3mg F6 CC-122 plus obinutuzumab in patients with R/R FL.
Abstract 039
Another phase IB dose escalation and expansion study was the topic of the next abstract presented by Connie Lee Batlevi from Memorial Sloan Kettering Cancer Center, New York, NY, USA. This study evaluated a combination of ibrutinib and buparlisib in patients with R/R FLBCL, MCL, and FL. Once again, the rationale for this study was pre-clinical data that has demonstrated synergism between BTK and PI3K inhibitors in B-cell NHL.

Patients were included in the study if they had:
- R/R DLBCL: more than one line of prior therapy and ASCT unless ineligible, transformed histologies permitted
- R/R MCL with more than one line of prior therapy
- R/R FL with more than two line of prior therapy
So far, 25 patients have enrolled (10 DLBCL, 5 FL, 10 MCL) with median number of prior systemic therapies being 4 for DLBCL (range, 1–7), 2 for FL (all had 2 prior regimens), and 1 for MCL (range, 1–2). Twenty-three patients completed at least one cycle and were evaluable for toxicity.
Patients were administered escalating doses of once daily ibrutinib and buparlisib in three dose levels (ibrutinib 420–560mg; buparlisib 80–100 mg). Dose level 3 (Ibrutinib 560mg, buparlisib 100mg) was selected for dose expansion based on 1/6 patients with DLT.
|
Dose Level |
N |
Ibrutinib (mg) |
Buparlisib (mg) |
DLT |
|---|---|---|---|---|
|
1 |
6 |
420 |
80 |
1/6 (grade 3 anorexia resulting in hospitalization) |
|
2 |
4 |
560 |
80 |
0/4 |
|
3 |
6 |
560 |
100 |
1/6 (grade 2 cerebrovascular infarct of pre-existing pituitary adenoma, patient taken off study) |
In the safety analysis (n=27), 63% of patients had a ≥grade 3 AE; the most frequent were hyperglycemia (19%), rash (19%), diarrhea (11%), anorexia (11%0, and neurologic changes (11%; included depression, agitation, mood swings, confusion, memory impairment). All neurologic changes resolved upon withholding buparlisib. Eighty-six percent of patients adhered to treatment with ibrutinib, this was only 64% with buparlisib.
Overall Response Rate for all patients (n=25) using the Lugano Response Criteria was 52% with a 40% CR rate. ORR specifically in DLBCL, FL, and MCL was 11%, 20%, and 100%, respectively.
Batlevi concluded the talk by stating that the combination of ibrutinib plus buparlisib resulted in manageably predicted toxicities for both the BTK and PI3K inhibitors. The combination also demonstrated encouraging clinical activity in patients with relapsed MCL. Enrolment for the phase IB expansion portions of the study are ongoing to more accurately evaluate the safety and efficacy of ibrutinib plus buparlisib.
Abstract 040
The last talk in this session was given by Matthew Davids, MD, MMSc, from the Dana-Farber Cancer Institute, Boston, MA, USA, and included updated results of another phase I/IB study of ibrutinib and TGR-1202 in patients with R/R MCL or CLL (NCT02268851).
R/R MCL and CLL patients’ achieve limited depth and durability in their responses to ibrutinib. TGR-1202 (umbralisib) is a novel oral PI3Kδ inhibitor which carries a more favorable toxicity profile than other PI3K inhibitors. The group hypothesized that combining PI3K/BTK blockade using TGR-1202 and ibrutinib would be a tolerable and efficacious strategy to treat patients with R/R MCL and CLL.
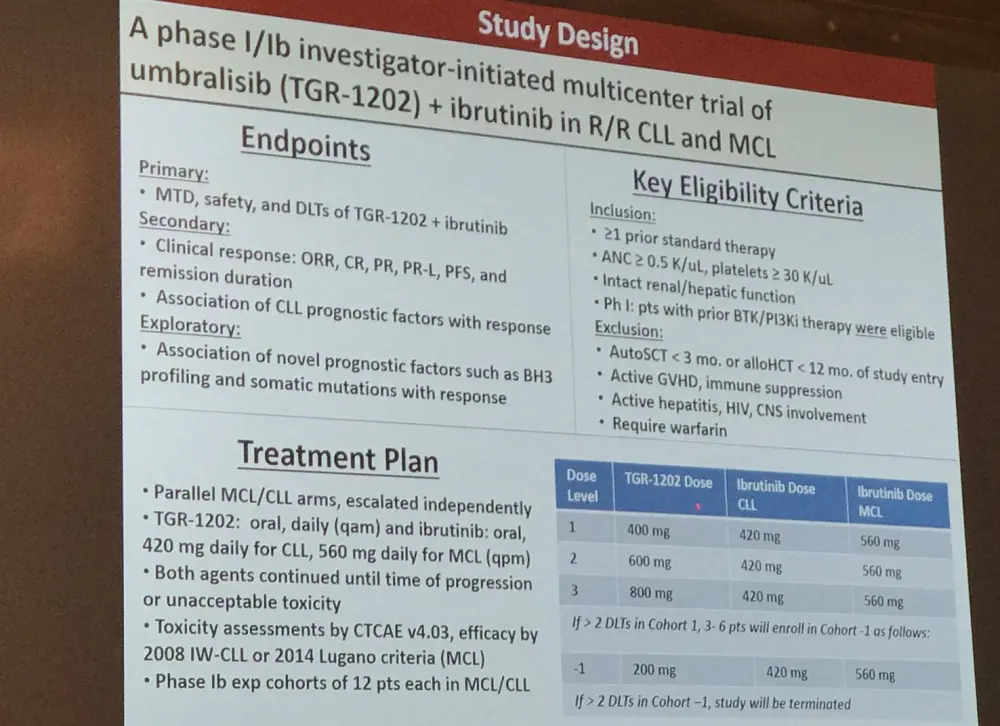
Overall, 33 patients have enrolled on the study, including 15 MCL and 18 CLL patients. The median age at enrollment was 67 years (range, 48–83) and the median number of prior therapies was 3 for MCL (range, 2–5; including 4 with prior ASCT) and 2 for CLL (range, 1–6). In CLL patients, del(17p) was detected in 4/17 (24%), del(11q) in 7/17 (41%), and unmutated IGHV in 11/17 (65%) patients.
In the phase I portion of the trial, no DLTs were observed but an MTD was not identified. The recommended phase II dose of TGR-1202 for both MCL and CLL was 800mg. The incidence of hematologic toxicities reported in the safety analysis are presented below:
|
|
CLL (n=18) |
MCL (n=14) |
|---|---|---|
|
Neutropenia |
38%; 17% grade 3–4 |
36%; 7.1% grade 3–4 |
|
Thrombocytopenia |
11% all grade 1 |
36%; 7.1% grade 3 |
|
Anemia |
15% all grade 1–2 |
29%; 7.1% grade 3 |
Other toxicities of special interest included:
- Diarrhea: 11/32 (34%) of patients (28% grade 1, 6% grade 2, no inflammatory colitis)
- Transaminitis: 7/32 (22%) of patients (all grade 1 and self-limited, no treatment interruption)
- Pneumonitis: 1/32 (3%), grade 1
- Bleeding events: grade 1 epistaxis, hematuria, vitreous hemorrhage in one CLL patient each
- Atrial fibrillation: 2/32 (6%), both grade 3
- Infection: 8/32 (25%), 4 grade 1–2; 2 grade 3 aspergillus, 1 c. diff, 1 grade 4 influenza
In CLL patients, the ORR was 94% (16/17) with a CR of 16% (1/17; 3 others with radiographic CR), and PR/PR-L in 88% (15/17). All three patients who had previously received a PI3K inhibitor, and one of two patients who had received previous ibrutinib, responded. In MCL patients, the ORR was 79% (11/14) with a CR of 9% (1/11; 1 other with radiographic CR) and a PR of 71% (10/11).
Median follow-up time among survivors was 14 months (range, 0.8–29.5). 1-year PFS for CLL is 88% and 1-year OS is 94%. Median PFS and OS for MCL is 8.4 and 11.6 months, respectively. One CLL patient has died, attributed to progressive disease; and 6 MCL patients have died (5 due to PD, 1 due to toxicity from next therapy).
In conclusion, the combination of TGR-1202 and ibrutinib was well tolerated in patients with R/R CLL and MCL, with no DLTs observed and a recommended phase II dose of TGR-1202 of 800mg. Toxicities were manageable. Preliminary efficacy data shows a high rate of response in either disease subtype. Correlative studies are currently in progress and enrolment continues in the MCL arm.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average vein-to-vein time when treating patients with DLBCL with a reimbursed CAR T-cell therapy (from apheresis to infusion)?

