All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
iwCLL 2017 | Epigenetics of CLL: understanding ontogeny and clinical behavior
At iwCLL 2017, on Monday 13th May, the second half of the session titled “Unraveling the factors leading to the development of CLL”, and was chaired by Emili Montserrat (Hospital Clinic of Barcelona) and John Gribben (Barts Cancer Institute).
During this session, a talk titled “Harnessing epigenetics to further our understanding of CLL ontogeny and clinical behavior” was presented by Christopher Oakes, PhD, from The Ohio State University Wexner Medical Center, Columbus, Ohio, US.
Oakes began the talk by explaining what epigenetics is: heritable information not encoded in the DNA sequence (A/T/C/G). Epigenetic states are programmed in cell lineage development and become fixed in mature, differentiated cells defining their cellular identity. Cancer cells acquire aberrant epigenetic marks that are key to a malignant phenotype.
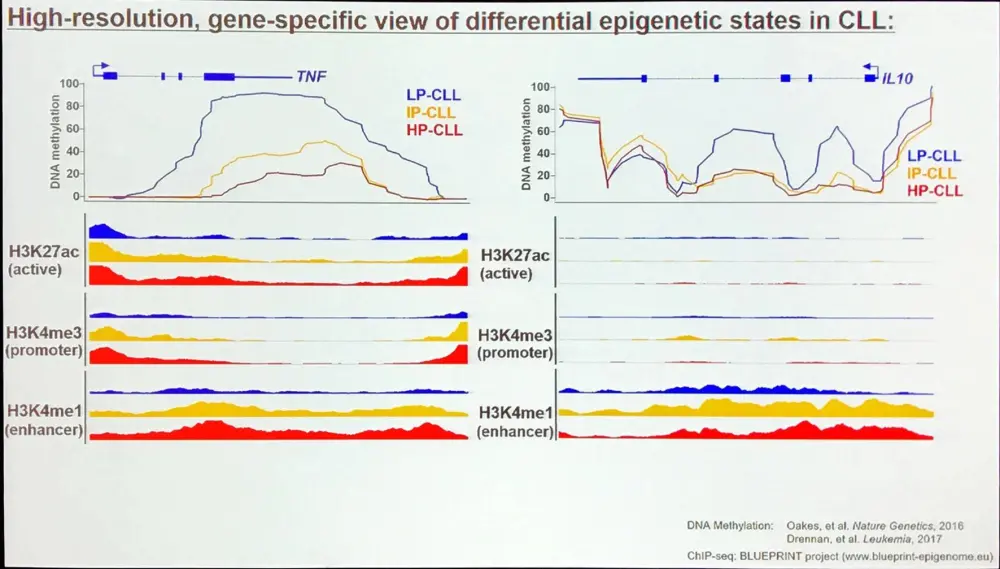
Where do the different epigenetic patterns in CLL come from?
CLL subgroup-specific methylation states are mirrored in normal B-cell subsets. A huge amount of DNA methylation takes place during maturation of B-cells and DNA methylation programming is associated with numerous annotated pathways:
|
Members of the BCR signaling pathway |
8.62 |
|---|---|
|
Genes related to PIP3 signaling in B-lymphocytes |
6.21 |
|
Chronic Myeloid Leukemia |
5.95 |
|
Gene regulation by peroxisome proliferators via PPARα |
5.93 |
|
B-Cell Antigen Receptor |
5.56 |
|
Trefoil Factors initiate mucosal healing |
4.96 |
|
Role of Erk5 in neuronal survival |
4.92 |
|
B-Cell survival pathway |
4.91 |
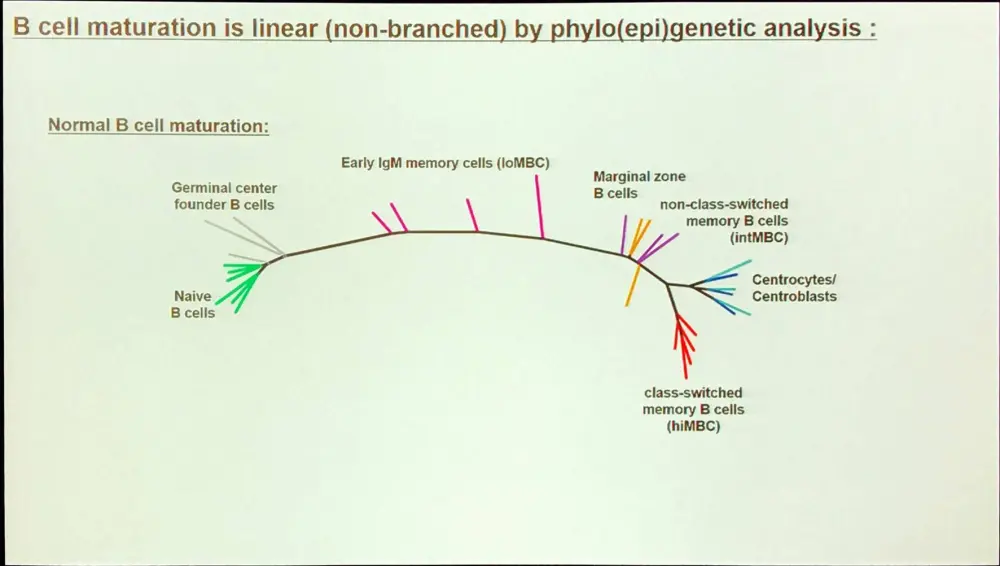
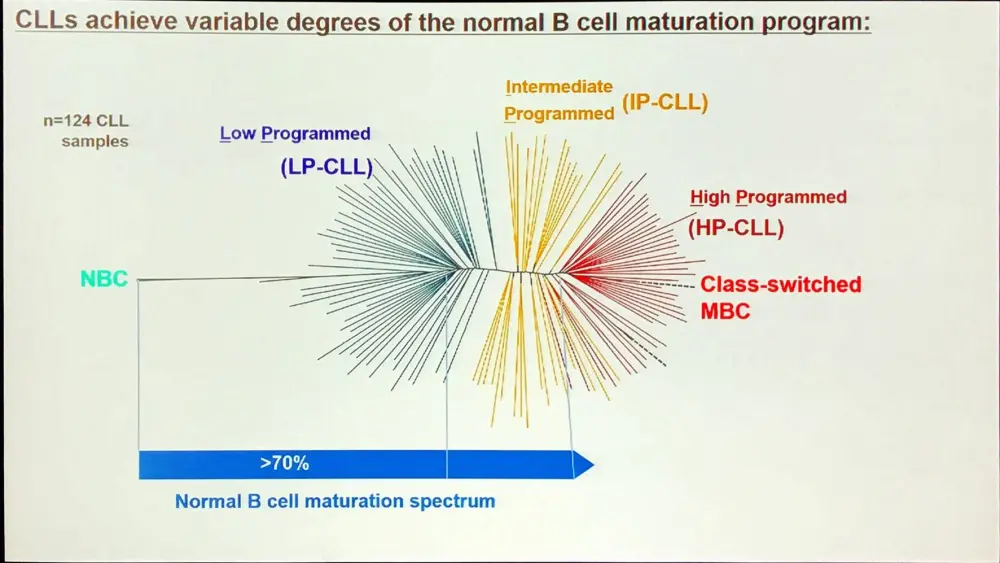
A large proportion of the epigenetic patterns observed in CLL derives from the epigenetic status of the founder cell.
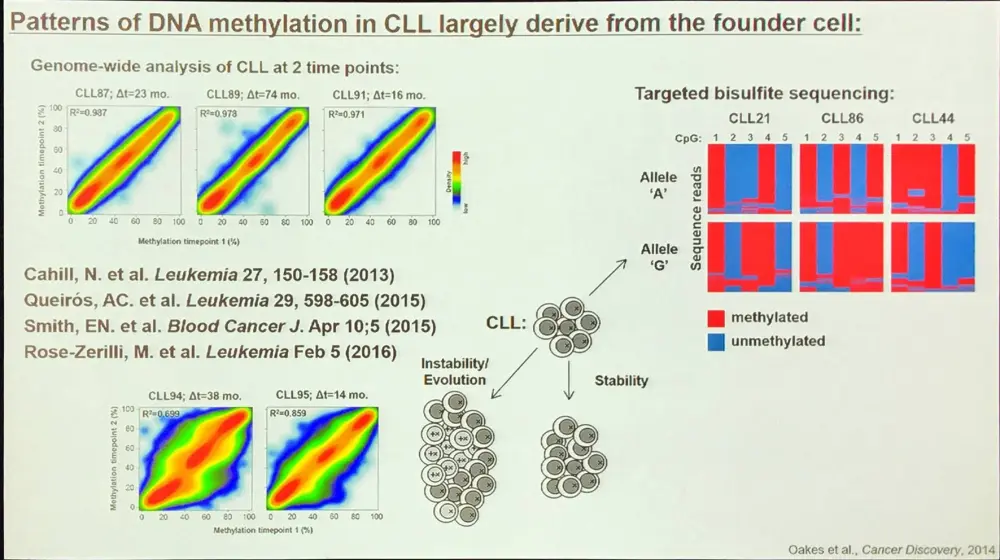
Additionally, aberrant DNA methylation programming of Low Programmed (LP)-CLL involves excess programming of NFAT and EGR as well as reduced programming of EBF and AP-1, unbalancing the normal B-cell epigenetic program (Oakes et al. 2016). CLL-specific methylation may involve aberrant BCR signaling.
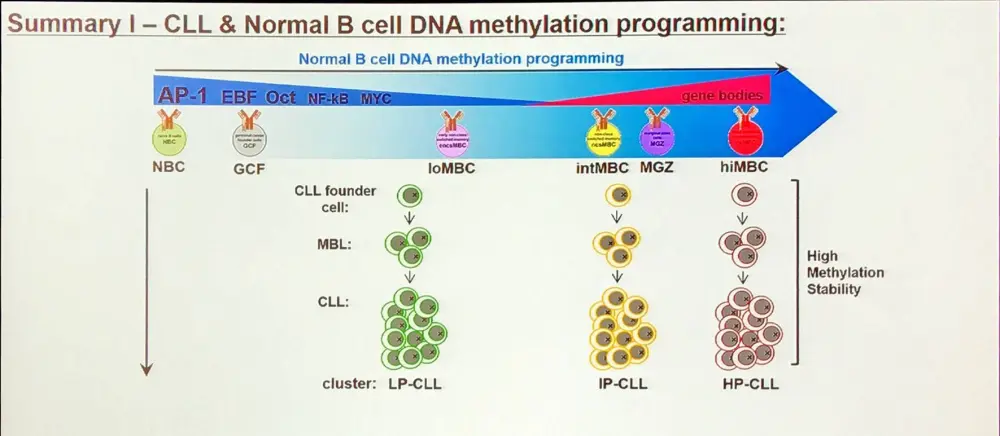
Overall, in CLL, the majority of methylation marks relate to normal B-cell maturation. Moreover, CLL methylation pattern is closer to memory than naïve B-cells. However, LP-CLL selectively maintain naïve B-cell states at specific genes. Lastly, the epitype derives from the maturation state of the cell of origin.
Do epigenetic patterns influence the biology of CLL?
Methylation states control gene expression in CLL (Drennan et al. 2017; Oakes et al. 2016); modulating the expression of hundreds of genes. Additionally, DNA methylation subgroups in CLL has a strong association with clinical outcomes.
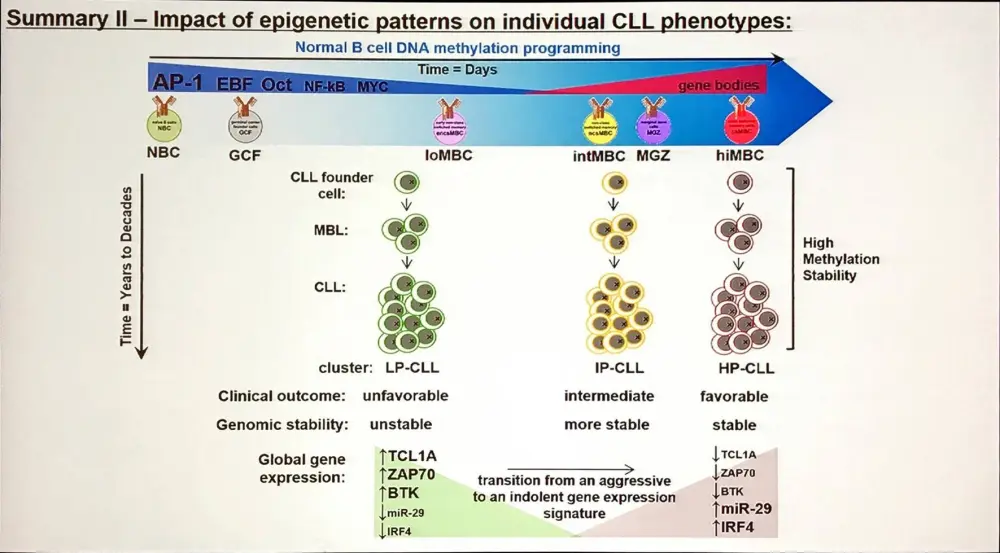
How can we make use of this information?
DNA methylation marks make for powerful biomarkers, for example methylation of ZAP-70:
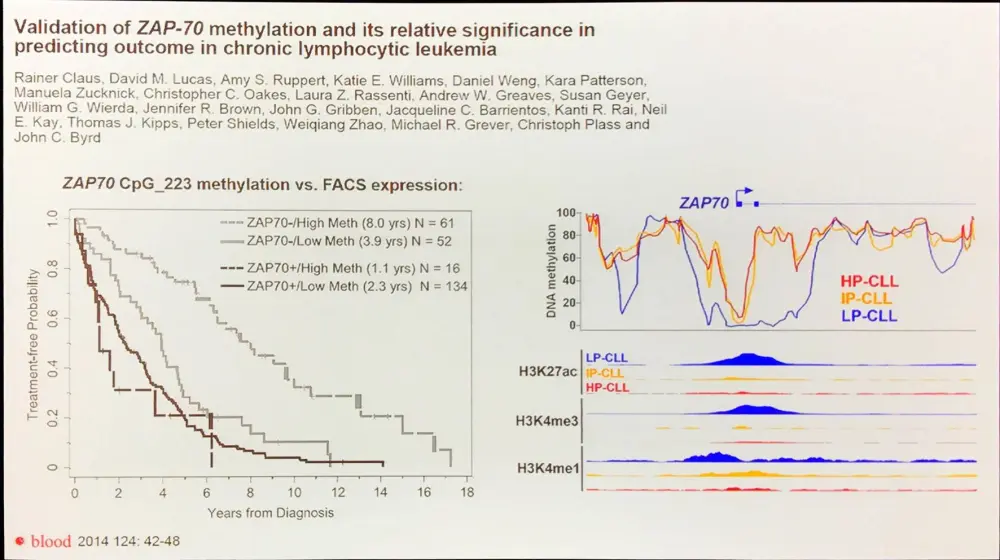
Epigenomic signatures may also reveal the cellular events surrounding the genesis of individual CLLs, identifying potential novel therapeutic vulnerabilities.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average time to secure a reimbursed CAR T-cell therapy manufacturing slot for patients with DLBCL (from decision to treatment with a CAR T-cell therapy)?

