All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
ICML 2017 | Molecular classification, outcomes, and treatment – Primary Mediastinal B-Cell Lymphoma Session
Bookmark this article
A session focused on Primary Mediastinal B-Cell Lymphoma (PMBCL) was held during the 14th International Conference on Malignant Lymphoma (ICML), Lugano, Switzerland, and was co-chaired by A. Davies (University of Southampton, UK) and G.S. Nowakowski (Mayo Clinic, Rochester, MN, US).
Abstract 047
The first talk, given by A. Mottok from the BC Cancer Agency, Vancouver, Canada, focused on using Formalin-Fixed Paraffin Embedded (FFPE) samples to molecularly classify PMBCLs.
PMBCL is acknowledged as a distinct category of Lymphoma in the current WHO classification and represents 2–4% of all B-cell Non-Hodgkin Lymphoma. Pathogenic hallmarks include JAK-STAT pathway, NF-kB pathway, and immune checkpoints.
Diagnosis of PMBCL is dependent on integration of clinical characteristics and presentation, as distinguishing it from Diffuse Large B-Cell Lymphoma (DLBCL) based on morphological or immunophenotypic features alone can be challenging. Moreover, clinico-pathologic consensus is not always well instituted.
Gene Expression Profiling (GEP) studies have demonstrated that PMBCL can be distinguished from DLBCL on a molecular level and supported a strong relationship between PMBCL and classical Hodgkin Lymphoma. However, these studies were performed using snap-frozen tissue and so the molecular classification of PMBCL has not yet been widely adopted into clinical practice. The group aimed to develop a robust and accurate molecular assay for the distinction of PMBCL from DLBCL based on gene expression measurements in routinely available FFPE biopsies.
In this study, all cases were centrally reviewed by a panel of expert hemato-pathologists in the Lymphoma and Leukemia Molecular Profiling Project consortium. Gene selection was based on previous data generated from over 300 cases on Affymetrix U133 plus 2.0 microarrays and the NanoString platform. The training cohort for the new Lymph3Cx assay was made up of 68 cases (48 DLBCL and 20 PMBCL); the independent validation cohort comprised 88 PMBCL and 78 DLBCL cases. Cases were required to have a tumor content of ≥60% and nucleic acids were extracted from 10mm FFPE scrolls. Digital gene expression was performed on 200ng RNA using NanoString technology.
Sixty-four probes, including the 20 genes previously reported in the Lymph2Cx assay, made up the final Lymph3Cx gene set. A probabilistic model accounting for classification error was trained to produce model scores that best distinguished between DLBCL and PMBCL. Cut-points were defined at the 0.1 and 0.9 probability scores. The model was applied to the independent validation cohort. Gene expression data of sufficient quality was acquired in 94.6% (157/166) of cases. Among pathologically-defined PMBCL cases, the molecular signature was in agreement in 85% of cases. Ten percent of cases were assigned to the “uncertain” category and 5% showed a molecular signature of DLBCL. Among pathologically-defined DLBCL cases, the assay classified 83% as DLBCL, 14% were “uncertain”, and 3% were predicted to be PMBCL.
|
Pathology review |
Predicted DLBCL |
Uncertain PMBCL/DLBCL |
Predicted PMBCL |
|---|---|---|---|
|
DLBCL |
60 (83%) |
10 (14%) |
2 (3%) |
|
PMBCL |
4 (5%) |
9 (10%) |
75 (85%) |
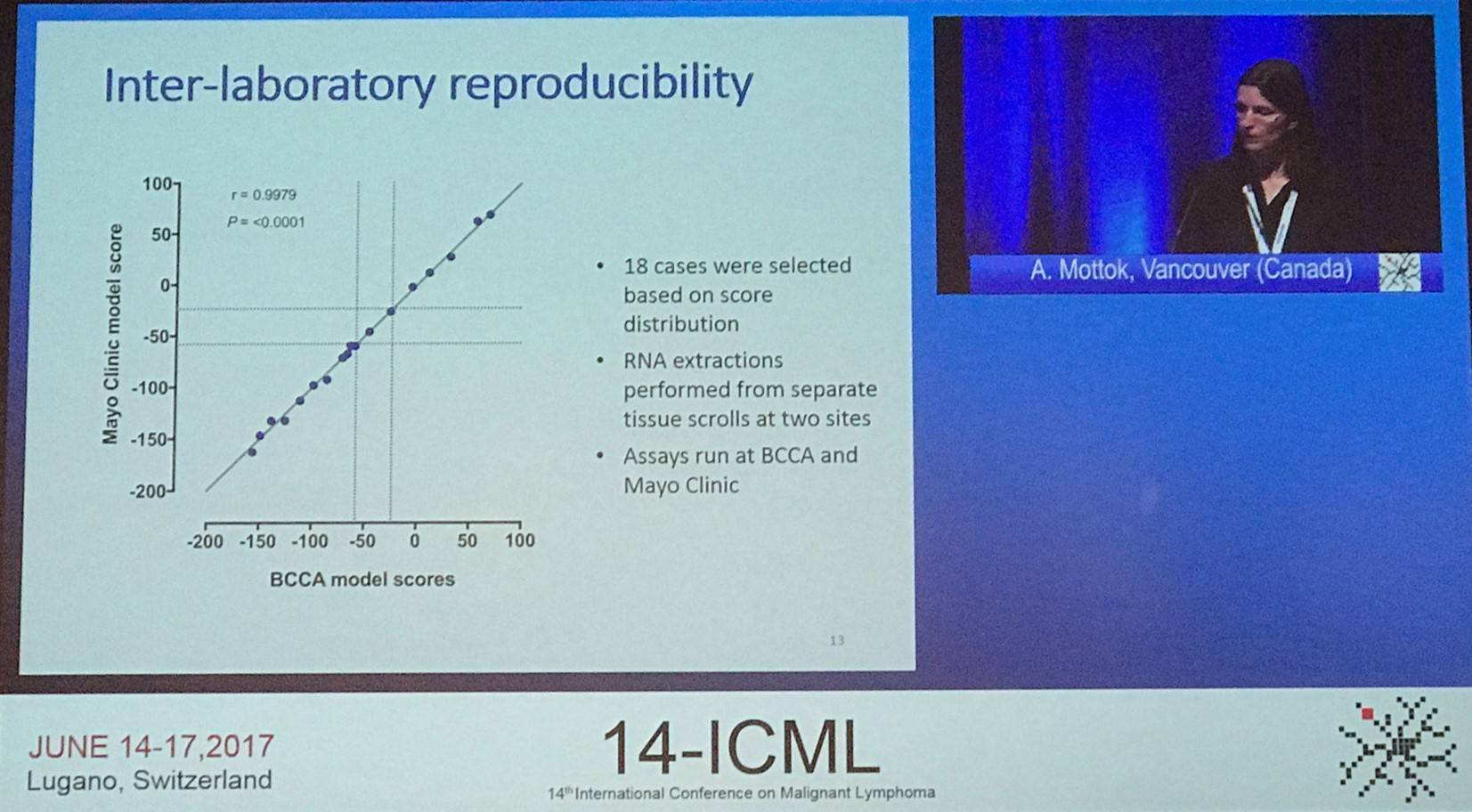
Mottock concluded the talk by stating that the new GEP-based assay to distinguish PMBCL and DLBCL based on FFPE samples is applicable. The molecular classification shows high concordance with the pathological classification of an expert hemato-pathologist panel. Future studies need to validate its utility for routine diagnostic purposes and therapeutic decision making.
Abstract 048
L. Ceriani from the Oncology Institute of Southern Switzerland, Bellinzona, Switzerland, presented the second abstract titled “Metabolic heterogeneity of baseline 18-FDG PET-CT scan predicts outcome in Primary Mediastinal B-Cell Lymphoma.”
Malignant tumors show intra-tumoral biological heterogeneity associated with cellular and molecular characteristics; for example, cellular proliferation, necrosis, fibrosis, changes in blood flow, angiogenesis, cellular metabolism, hypoxia, and expression of particular receptors. High intra-tumoral heterogeneity appears prognostic and may predict treatment failure.
18F-Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography (18-FDG PET-CT) has been suggested as a reliable tool in order to evaluate the metabolic patterns of intra-tumoral heterogeneity. Preliminary results have been promising for Metabolic Heterogeneity (MH) measured on baseline 18-FDG PET-CT in predicting outcomes of sarcoma/solid tumor patients.
The study presented by Ceriani aimed to evaluate if MH of mediastinal masses, alone and/or in association with other baseline quantitative PET parameters, may potentially be a prognostic marker in PMBCL patients.
MH was estimated in a cohort of 103 PMBCL patients enrolled in the IELSG-26 trial using 18-FDG PET-CT. All patients had been administered combination chemoimmunotherapy with doxorubicin and rituximab-based regimens. Of these, 93 had also received consolidation radiotherapy. Median follow-up was 60 months, and 13 progression of disease and 6 deaths were registered. Mediastinal masses were segmented by means of a fixed threshold algorithm with a 25% SUVmax cut-off. MH was measured by the estimation of the Area Under Curve (AUC) of Cumulative SUV-volume histogram (AUC-CSH) and the calculation of the Coefficient of Variation (COV) of the SUVmax values.
Correlation between the two methods was good (r = -0.073; P < 0.0001). MH did not significantly associate with other quantitative PET-derived parameters (SUVmax, MTV, and TLG); however, a weak association (r = 0.2) was found with MTV when MH was measured as AUC-CSH. Although, when MH was measured as COV, this correlation disappeared (r = 0.07).
Both approaches resulted in “superimposable results” when the impact of MH on treatment outcome was assessed. At 5 years, Overall Survival (OS) was 96% for patients with low AUC-CSH heterogeneity compared to 50% for those with high heterogeneity (P < 0.0001) while Progression-Free Survival (PFS) was 94% compared to 73%, respectively (P = 0.0001). In univariate analysis, elevated MH was significantly associated with worse PFS and OS. Increased MH identified patients with worse prognosis with high NPV but low PPV.
In Cox models including MH, MTV, TLG, and tumor bulk (> 10 cm), TLG (P = 0.0002) and MH (p = 0.0002) retained statistical significance in PFS analysis, and MH was the only predictor of shorter OS (P = 0.019). The association of high MH and high TLG identified progressive patients (PPV = 89%) improving the performance of each single parameter (without negatively effecting NPV = 95%).
|
Parameter |
Positive predictive value for PFS |
Negative predictive value for PFS |
||
|---|---|---|---|---|
|
MH |
9/34 |
26% |
65/69 |
94% |
|
TLG baseline |
12/33 |
36% |
69/70 |
99% |
|
TLG + MH (both high) |
8/9 |
89% |
89/94 |
95% |

Ceriani concluded the talk by stating that MH on baseline PET seems to be “a powerful predictor of PMBCL outcomes and warrant further validation as a biomarker.” Moreover, a combined model of TLH and MH could potentially identify patients with worse outcomes earlier.
Abstract 049
The third talk in this session was focused on a multicenter, retrospective analysis investigating the outcomes of patients of different age ranges who had received Dose-Adjusted (DA) EPOCH-R, and was given by Lisa Giulino-Roth, MD, from Weill Cornell Medicine, New York, NY, US.
Roth began with an overview of the presentation and management of PMBCL. Clinical presentation occurs in both children and adults, peaking in the Adolescents and Young Adults (AYA) age range (15–39 years). PMBCL is more common in females and usually presents as a large mediastinal mass. Currently, there is no single standard of care, but DA-EPOCH-R has been adopted as the standard in numerous centers despite a lack of data in the multi-cancer setting.
Following this, results from some single-center experiences of DA-EPOCH-R without Radiotherapy (RT) in adult PMBCL patients were outlined:
|
Center |
Study type |
n |
Age median (range) |
Median follow-up |
EFS |
|---|---|---|---|---|---|
|
Prospective Phase II |
51 |
30y (19–52) |
63 months |
93% |
|
|
Retrospective |
16 |
33y (22–68) |
37 months |
100% |
|
|
Retrospective |
25 |
35y (19–70) |
NR |
~85% |
The current retrospective study included 156 PMBCL patients treated with DA-EPOCH-R from 24 different academic medical centers, including 38 pediatric and 118 adult patients. The objectives of the analysis were:
- To determine outcomes in a multicenter setting
- Investigate potential differences between pediatric and adult patients
- Evaluate the predictive value of end-of-therapy FDG-PET
Following this, the patient characteristics were outlined; bulky mediastinal disease was more common in pediatric than adult patients and pediatric patients were more likely to be escalated to at least dose level 4.
|
|
Total cohort n=156 |
Pediatric (age <21) n=38 |
Adult (age ≥21) n=118 |
P value ped vs. adult |
|
|---|---|---|---|---|---|
|
Age in years (range) |
31 (9–70) |
16 (9–20) |
34 (21–70) |
<0.01 |
|
|
Female (%) |
64% |
55% |
67% |
0.243 |
|
|
ECOG PS median |
1 |
N/A |
1 |
N/A |
|
|
Stage |
I |
17% |
3% |
21% |
N/A |
|
II |
44% |
24% |
51% |
||
|
III |
19% |
60% |
6% |
||
|
IV |
20% |
13% |
22% |
||
|
Bulky tumor >10cm (%) |
63% |
78% |
58% |
0.031 |
|
|
LDH > ULN (%) |
83% |
86% |
82% |
0.799 |
|
|
B symptoms (%) |
40% |
31% |
43% |
0.244 |
|
|
Extranodal disease |
33% |
40% |
31% |
0.328 |
|
The incidence of thrombotic events was also discussed in detail. Overall, 28% of patients experienced thrombosis: upper extremity (n=22), internal jugular vein or SVC (n=10), intracardiac (n=5), PE (n=5), and lower extremity (n=2). Catheter-associated thrombosis was reported in 10% of patients; the rate was similar in PICC line (13%) versus mediport (9%; P = 0.565). A higher rate of thrombosis was found in pediatric compared to adult patients (46% vs. 23%; P = 0.011). Moreover, pediatric patients were more likely to undergo an echocardiogram (95% vs. 48%; P < 0.001).
The median follow-up was 22.6 months (range, 2.7–101.0 months), and 22 patients of the 156 experienced an event: relapse (n=9), primary refractory disease (n=2), death in a patient in remission, complication for T-E fistula repair (n=1). The median time from diagnosis to relapse was 6.0 months (2.8–14.8 months). Sites of relapse included the mediastinum with/without the lung (n=11), CNS (n=4), and multiple nodal/extranodal site (n=5). Of the 21 patients with Relapsed/Refractory (R/R) disease: 10 are alive and disease free, 6 alive with evidence of disease, and 5 died due to PMBCL.
It was found that outcomes between pediatric and adult patients were similar. 3-year EFS for the total cohort was 85.9%, and for pediatric and adult patients was 81.0% versus 87.4%, respectively (P = 0.338). 3-year OS for the entire cohort was 95.4% and for pediatric and adult patients was 90.7% versus 97.1%, respectively (P = 0.170).
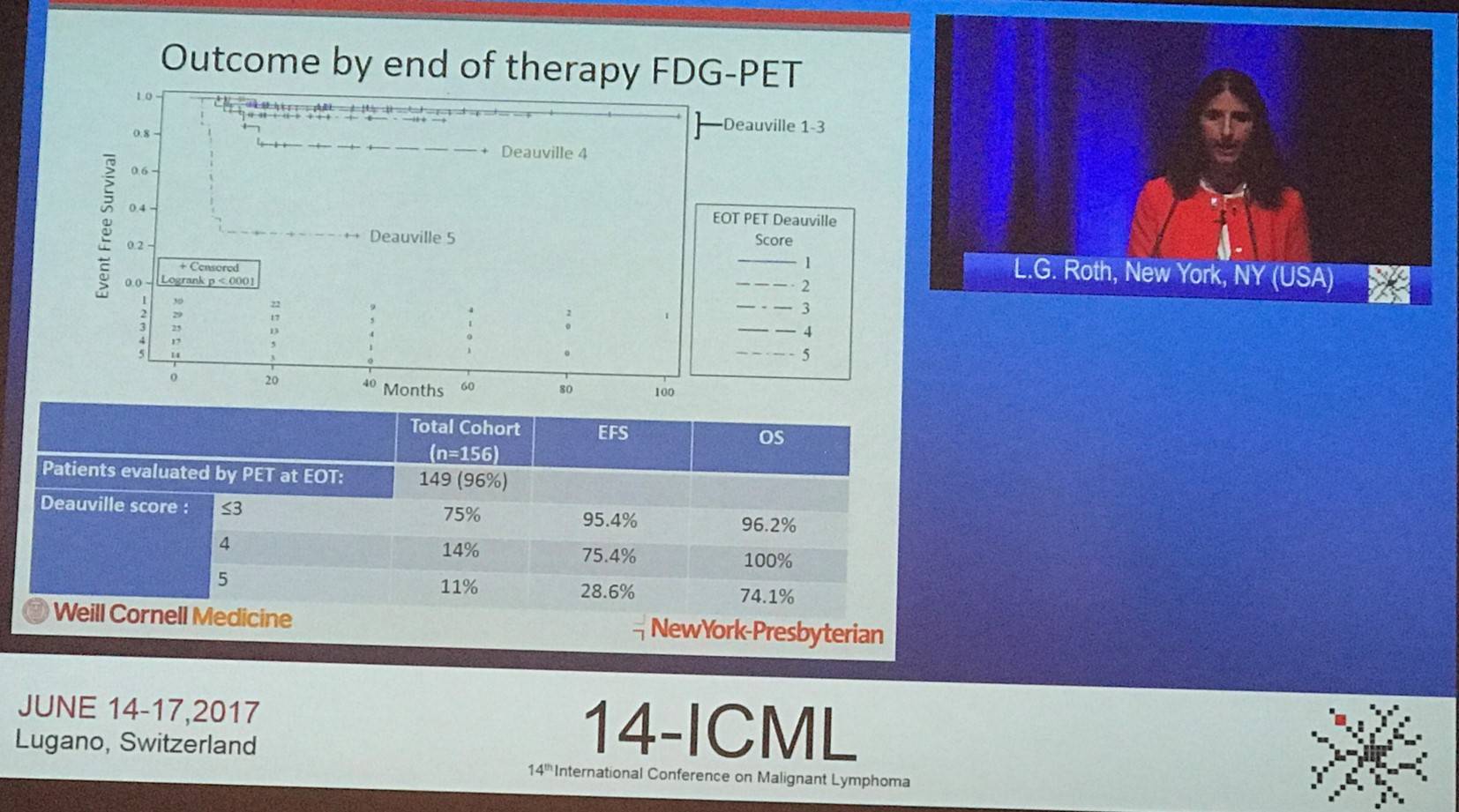
On completion of DA-EPOCH-R, three-quarters (75%) of patients had a negative FDG-PET scan (defined as Deauville score 1–3). Negative FDG-PET at end-of-therapy was associated with improved EFS (95.4% vs. 54.9%; P < 0.001).
Roth concluded this abstract presentation by stating that, to the group’s knowledge, this is the largest analysis of DA-EPOCH-R in PMBCL and the first report in pediatric patients. There were some limitations with the study including its retrospective nature, reliance of local pathology and radiology reviews, and RT was given at the discretion of the treating physician. The results in this retrospective analysis support the use of DA-EPOCH-R for the treatment of pediatric, adolescent, and adult patients with PMBCL. It was observed that a high rate of thrombosis occurs with this treatment, and therefore prophylactic anti-coagulation should be considered. Additionally, patients who are PET-positive at the end of therapy were found to have worse outcomes and so it may be beneficial for these patients to receive augmented or novel therapies.
Abstract 050
Pier Luigi Zinzani, MD, PhD, from the University of Bologna, Bologna, Italy, presented the final talk in this session which conveyed interim analysis of the phase II KEYNOTE-170 trial of pembrolizumab for R/R PMBCL (NCT02576990).
Standard front-line therapy cures 80% of patients with PMBCL. However, approximately 200 patients per year are diagnosed with R/R PMBCL which has a poor prognosis and has limited treatment options. New therapeutic options are needed, but due to the small numbers of patients hampers the ability to conduct clinical trials into novel agents.
Amplifications/translocations of 9p24.1 resulting in PD-L1 and/or PD-L2 overexpression are often observed in patients with PMBCL. Pembrolizumab is an anti-PD-1 antibody inhibits PD-1 binding to its ligands, PD-L1 and PD-L2, resulting in the restoration of tumor immune surveillance. In a phase I trial, pembrolizumab achieve an ORR of 48% and a CR of 24% in patients with R/R PMBCL (Zinzani et al. ICML. 2017. Poster #190).
The KEYNOTE-170 trial has a primary outcome measure of ORR, assessed by PET and CT scans per 2007 IWG response criteria by blinded central review. Secondary endpoints include DoR, PFS, OS, and safety.

Adult patients were eligible if they had R/R PMBCL and failed or are ineligible for ASCT (ineligible for ASCT must have failed two or more lines of previous treatment). Patients received pembrolizumab 200mg IV every 3 weeks until disease progression, unacceptable toxicity, or completion of 35 treatment cycles.
The study enrolled patients from 14 different centers across 9 countries. The efficacy population included all patients that had been administered one or more doses of the study drug and expected to have 24 weeks or more of follow-up after the analysis cut-off date. The safety population included all patients who had received one or more doses of study drug.
|
Safety population (n=49) |
||
|---|---|---|
|
Age, median (range) |
33 years (20–61) |
|
|
Female, n |
27 (55%) |
|
|
ECOG PS, n |
0 |
21 (43%) |
|
1 |
28 (57%) |
|
|
No prior transplant |
36 (74%) |
|
|
Prior lines of therapy |
Median (range) |
3 (2–8) |
|
≥2, n |
49 (100%) |
|
|
Prior radiation, n |
15 (31%) |
|
|
Prior rituximab, n |
49 (100%) |
|
Median follow-up was 6.6 months (0.1–13.6) and 10 responses were ongoing at the time of analysis. The median time to response was 2.9 months (2.4–8.5) and median response duration was not reached (1.1–8.2 months). Best response by blinded central review was presented in a table by Zinzani:
|
Efficacy population (n=29) |
n |
% (95% CI) |
|---|---|---|
|
Overall Response |
12 |
41% (24–61) |
|
Complete Response |
4 |
14% (4–32) |
|
Partial Response |
8 |
28% (13–47) |
|
Stable Disease |
3 |
10% (2–27) |
|
Progressive Disease |
8 |
28% (13–47) |
|
No assessment |
6 |
21% (8–49) |
In patients who had failed ASCT (6/10), three patients (30%; 95% CI, 7–65) had a best response of CR, and a further three patients (30%; 95% CI, 7–65) had a best response of PR. In patients who were ineligible for ASCT (6/19), a best response of CR was achieved in one patient (5%; 95% CI, 0–26), and a best response of PR was achieved in five patients (25%; 95% CI, 9–51).
Zinzani also gave a summary of Adverse Events (AEs):
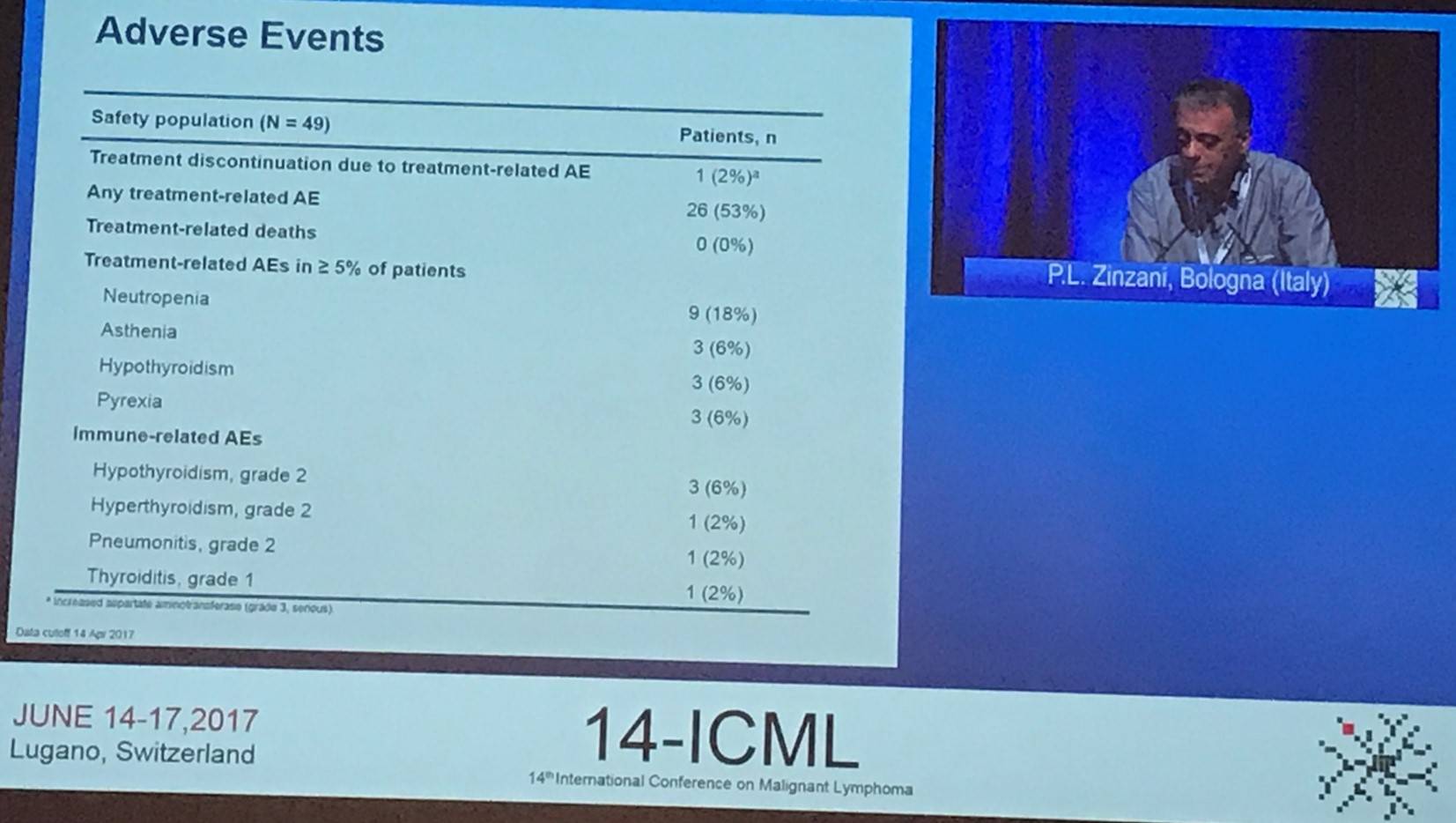
This abstract presentation was concluded by stating that pembrolizumab demonstrated encouraging anti-tumor activity and a manageable safety profile in patients with R/R PMBCL (including heavily pre-treated patients). These results are similar to that seen in the phase Ib KEYNOTE-013 trial (Zinzani et al. ICML. 2017. Poster #190). Enrolment is still ongoing, with a target of 53 patients in this cohort.
- Davies A. & Nowakowski G.S. Session 2 – Primary Mediastinal B-Cell Lymphoma. 14th International Conference on Malignant Lymphoma; 2017 June 14–17; Lugano, Switzerland.
- Mottock A. et al. Molecular classification of Primary Mediastinal Large B-Cell Lymphoma using Formalin-Fixed, Paraffin-Embedded tissue specimens – an LLMPP project. Hematological Oncology. 2017 June 07;35(suppl S2):59–60. DOI: 10.1002/hon.2437_46.
- Ceriani L. et al. Metabolic heterogeneity of baseline 18-FDG PET-CT scan predicts outcome in Primary Mediastinal B-Cell Lymphoma. Hematological Oncology. 2017 June 07;35(suppl S2):60–1. DOI: 10.1002/hon.2437_47.
- Roth L.G. et al. Outcomes of adults, adolescents, and children with Primary Mediastinal B-Cell Lymphoma treated with dose-adjusted EPOCH-R therapy: a multicenter retrospective analysis. Hematological Oncology. 2017 June 07;35(suppl S2):61–2. DOI: 10.1002/hon.2437_48.
- Zinani P.L. et al. Efficacy and safety of pembrolizumab in relapsed/refractory primary mediastinal large B-cell lymphoma (rrPMBCL): interim analysis of the KEYNOTE-170 phase 2 trial. Hematological Oncology. 2017 June 07;35(suppl S2):62–3. DOI: 10.1002/hon.2437_49.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








