All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a Healthcare Professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub uses cookies on this website. They help us give you the best online experience. By continuing to use our website without changing your cookie settings, you agree to our use of cookies in accordance with our updated Cookie Policy
Introducing

Now you can personalise
your Lymphoma Hub experience!
Bookmark content to read later
Select your specific areas of interest
View content recommended for you
Find out moreThe Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
iwCLL 2017 | Monocytes and Nurse-Like Cells have immunosuppressive features and support CLL cell survival
Bookmark this article
The second session at this year’s iwCLL was titled “Role of Non-Leukemic Cells and the Microenvironment in CLL Development”, and was jointly chaired by Silvia Deaglio (University of Turin, Torino, Italy) and Christopher Pepper (Cardiff University, Wales, UK).
During this session, Martina Seiffert, PhD, from the German Cancer Research Center, Heidelberg, Germany, gave a talk titled “From Monocytes to Nurse-Like Cells: Myeloid Cells Matter in CLL.”
The talk began by giving an update to the hallmarks of cancer; four “next generation” properties:
- Deregulating cellular energetics
- Genome instability and mutation
- Avoiding immune destruction
- Tumor-promoting inflammation
Tumor Associated Macrophages (TAMs) are the “undisputed stars of the inflammatory tumor microenvironment”:
- Decrease apoptosis and increase proliferation of tumor cells
- Increase angiogenesis
- Remodel the matrix
- Encourage metastasis (intravasation)
- Suppress the immune response toward the tumor
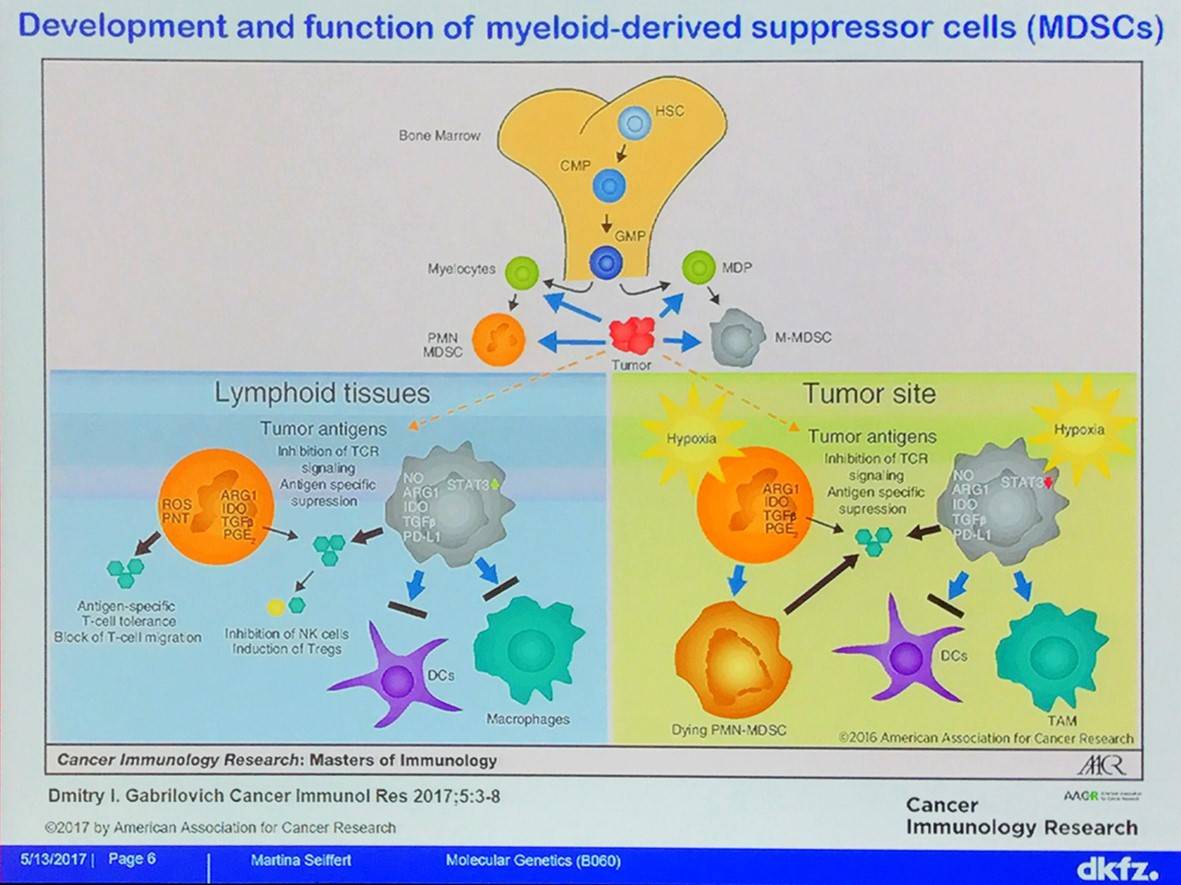
CLL monocytes and Nurselike Cells (NLCs) have immunosuppressive features; for example, they decrease effector T-cell proliferation and function as well as decrease phagocytosis by macrophages. NLCs have characteristics similar to that of TAMs. Monocytes and NLCs support survival of CLL cells in vitro.
Cytokine antibody arrays have detected 174 human or 144 murine serum proteins including CCL2, Axl, CXCL13, IL-1ra, L-Selectin and E-Selectin (P-Selectin in mice), and MMP1 (MMP9 in mice).
CLL is associated with a buildup of patrolling monocytes. In TCL1 mice, peritoneal macrophages are skewed towards a M2-like or TAM phenotype. They exhibit M2-like marker expression and signaling (CD206, IL-4Ra, CD86, PD-L2, p-STAT3, and p-STAT6). TCL1 monocytes have an inflammatory and immunosuppressive phenotype: express TREM-1 (regulates inflammatory responses and stimulates cytokine release) and PD-L1 (inhibitory co-stimulator, up-regulated on tumor cells and inhibits T-cell activity).
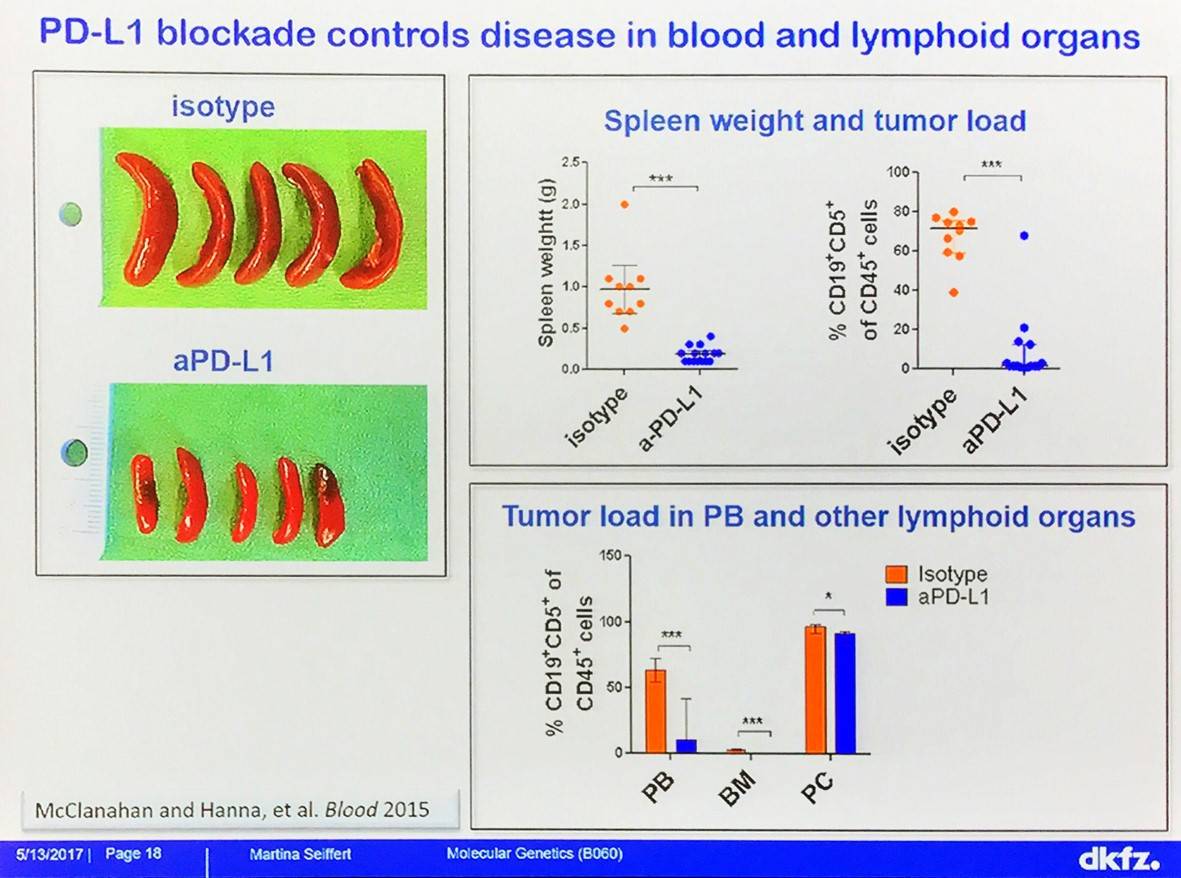
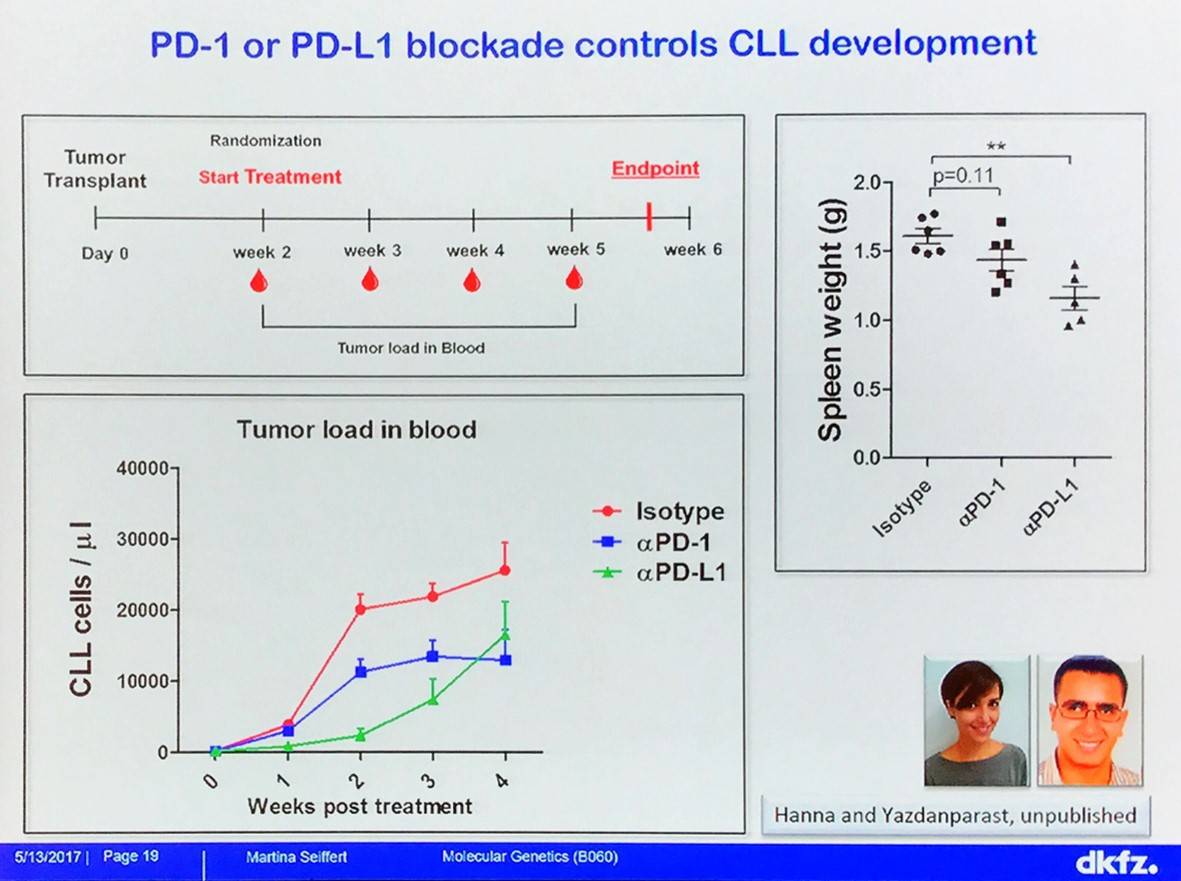
The effect of immune checkpoint blockade with anti-PD-L1 treatment on CLL has been evaluated in TCL1 mice:
Martina Seiffert then asked what induces the tumor-supportive phenotype in myeloid cells in CLL? CLL exosomes were isolated by serial centrifugation and verified by Electron Microscopy (EM) and immune-gold EM (particle size: 30–350 nm). Uptake of CLL exosomes induces PD-L1 expression and cytokine secretion in monocytes.
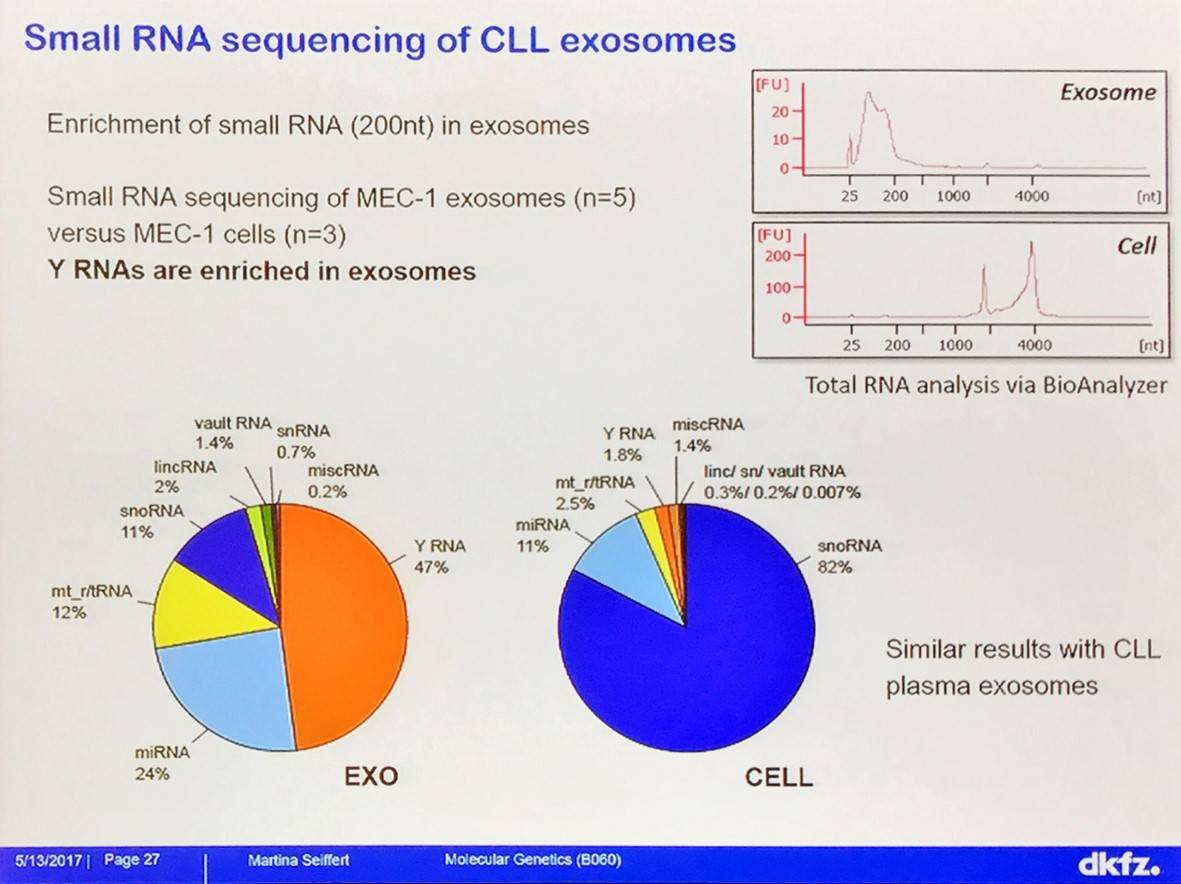
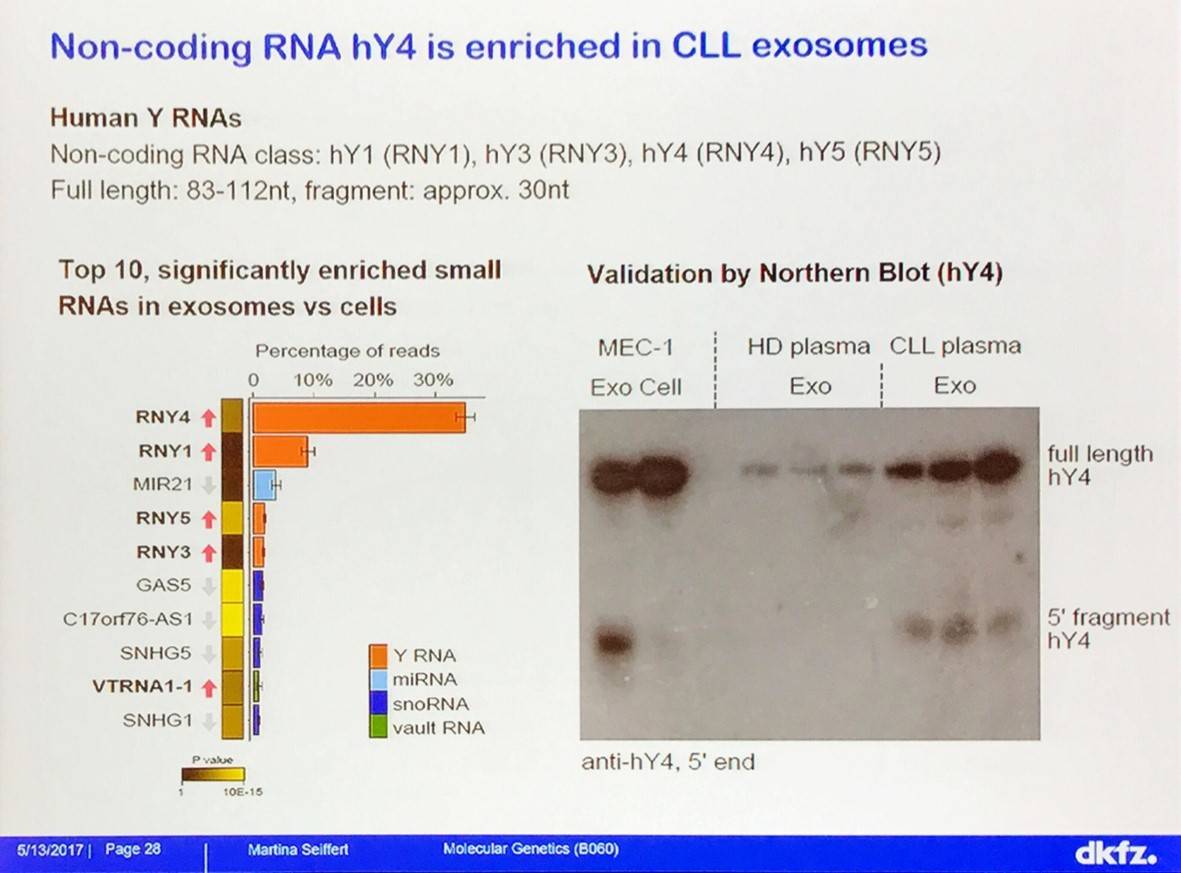
Uptake of Y RNA hY4 induces PD-L1 expression, cytokine secretion, and inflammatory response by monocytes:
|
Increase chemokines |
CCL2, CCL4, CCL5, CXCL9, CXCL10, CXCL11 |
|---|---|
|
Increase pro-inflammatory genes |
IL6, IL12A, TNF |
|
Increase immunosuppressive factors |
CD274 (PD-L1), IDO1, PDCD1 (PD-1) |
|
Decrease chemokine receptors |
CCR2, CXCR4 |
Many of these genes are deregulated in CLL. Response to hY4 in monocytes is TLR7-dependent; hY4 effects are abolished in TLR7 knock-out, but not MAVS knock-out or wild type mice.
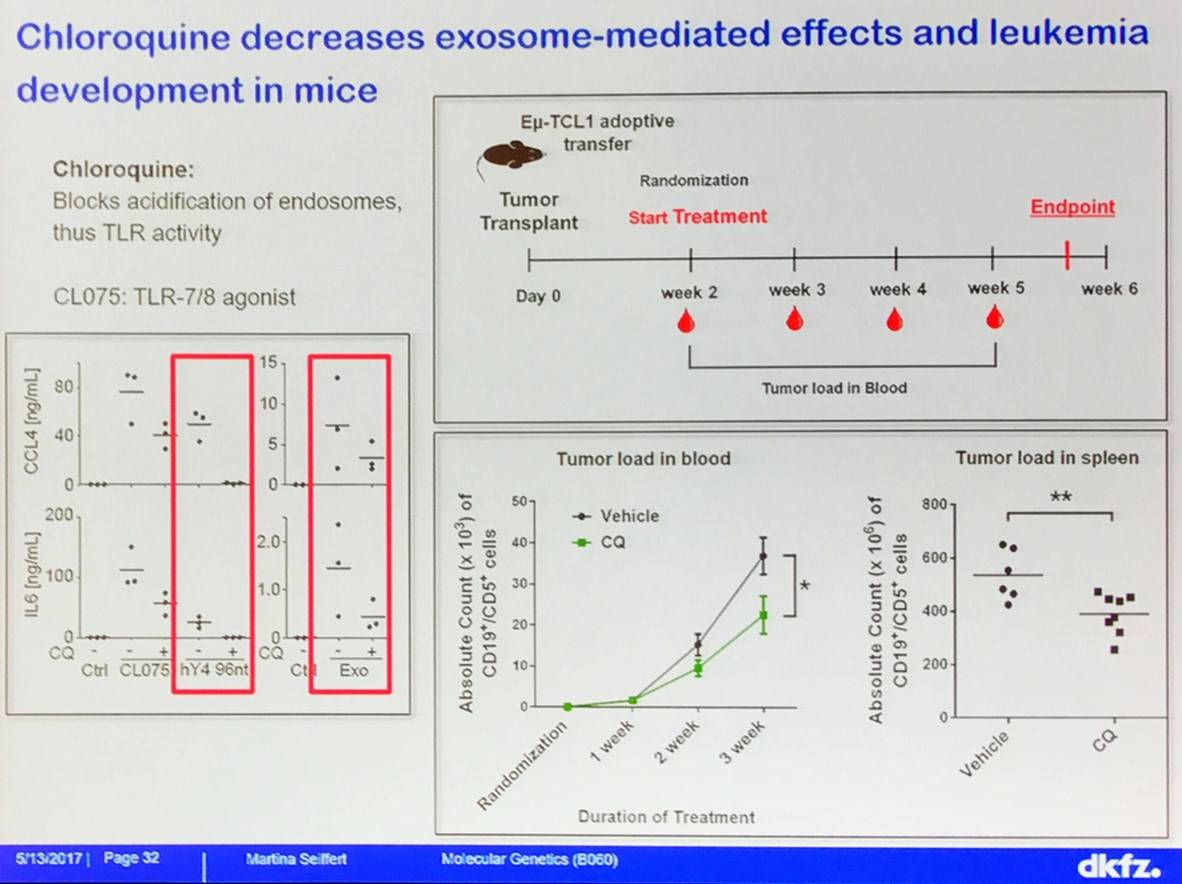
Martina Seiffert concluded the talk by outlining a novel proposed mechanism of myeloid phenotype induction in CLL:
- Y RNA are highly enriched in CLL exosomes, which are taken up by monocytes and macrophages
- Exosomes and Y RNA induce PD-L1 expression and cytokine secretion
- Y RNA-mediated response is dependent on endosomal TLR7
- Chloroquine decreases exosome-mediated effects on leukemia development in mice
- Seiffert M. From Monocytes to Nurse-Like Cells: Myeloid Cells Matter in CLL. XVII International Workshop on Chronic Lymphocytic Leukemia; 2017 May 12–15; New York, USA.

Understanding your specialty helps us to deliver the most relevant and engaging content.
Please spare a moment to share yours.
Please select or type your specialty
 Thank you
Thank youRelated articles
Newsletter
Subscribe to get the best content related to lymphoma & CLL delivered to your inbox








