All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Results from the OAsis phase I/II trial on ibrutinib, venetoclax plus obinutuzumab in patients with newly diagnosed mantle cell lymphoma
During the 2020 European Hematology Association (EHA) Annual Congress, Steven Le Gouill presented data from Cohort C of the OAsis phase I/II study (NCT02558816), investigating the safety and efficacy of ibrutinib, venetoclax plus obinutuzumab in patients with newly diagnosed mantle cell lymphoma (MCL).
The aim of this non-randomized trial was to identify the maximum tolerated dose of venetoclax when combined to fixed doses of ibrutinib and obinutuzumab. Secondary endpoints included safety, efficacy, response rates, progression-free survival, and overall survival.
Study design
The study included three cohorts:
- Cohort A (n = 9): patients with relapsed/refractory (R/R) MCL receiving ibrutinib and obinutuzumab
- Cohort B (n = 24): patients with R/R MCL receiving ibrutinib, venetoclax plus obinutuzumab
- Cohort C (n = 15): patients with newly diagnosed untreated MCL receiving ibrutinib, venetoclax plus obinutuzumab
- The treatment schedule is reported in Figure 1
Inclusion criteria for cohort C:
- Age ≥ 18 for French patients, age ≥ 16 for English patients
- Untreated patients with histologically confirmed MCL in need of treatment
- Disease stage II─IV
- Eastern Cooperative Oncology Group Performance Status 0─2
Figure 1. Treatment schedule1
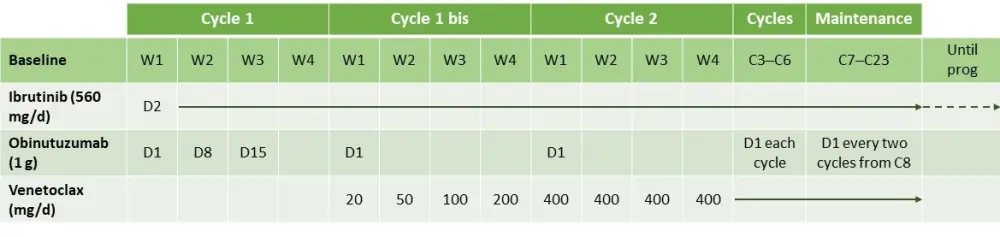
C, cycle; D, day; W, week
Patient characteristics
Patient characteristics are reported in Table 1. Patients were rather young with quite an aggressive disease.
Table 1. Patient characteristics1
|
Characteristic |
Patients (n = 15) |
|---|---|
|
BM, bone marrow; ECOG PS, Eastern Cooperative Oncology Group Performance Status; IGHV, immunoglobulin heavy chain variable region; MIPI, mantle cell lymphoma International Prognostic Index; NGS, next-generation sequencing; TP53, tumor protein 53 |
|
|
Median age, years (range) |
65 (51─77) |
|
Female/Male, n (%) |
9 (60)/ 6 (40) |
|
Stage III–IV, n (%) |
15 (100) |
|
BM involvement, n (%) |
8 (53) |
|
Pleomorphic, n (%) |
1 (7) |
|
Tumor size ˃ 5 cm, n (%) |
6 (40) |
|
ECOG PS < 2, n (%) |
15 (100) |
|
MIPI high risk, n (%) |
4 (27) |
|
MIPI intermediate risk, n (%) |
11 (73) |
|
Cytogenetic and molecular features, n (%) TP53 mutated (NGS) 17p deletion IGHV mutated (NGS) |
2 (13) 6 (40) 2 (13) |
Results
Safety
The triplet was well tolerated in patients who have never been exposed before to chemotherapeutic agents. The most common Grade 3 and 4 adverse events/serious adverse events, observed from Cycle 1 to Cycle 6, are reported in Table 2.
Table 2. Adverse events and serious adverse events from Cycle 1 to Cycle 61
|
ALAT, alanine aminotransferase; ASAT, aspartate aminotransferase |
||
|
Events |
Grade 3 |
Grade 4 |
|---|---|---|
|
Adverse events, n (%) All adverse events Neutropenia Lymphopenia Rash ALAT increased ASAT increased Hepatic cytolysis Hyperlymphocytosis |
5 (33) 2 (13) 1 (7) 1 (7) 0 1 (7) 0 1 (7) |
3 (20) 1 (7) 0 0 1 (7) 0 1 (7) 0 |
|
Serious adverse events All serious adverse events Appendicitis |
1 (7) 1 (7) |
0 0 |
Efficacy
The triplet induced very high complete remission (CR)/unconfirmed CR (CRu) rate very early. According to Cheson et al. (1999) criteria:2
- After Cycle 2
- Eight patients (53%) were in CR/CRu and seven (47%) in partial remission (PR)
- After Cycle 4
- 12 patients (80%) were in CR/CRu and two (13%) in PR
- After Cycle 6
- 12 patients (80%) were in CR/CRu and two (13%) in PR
According to the Lugano (2014) criteria:3
- After Cycle 6
- 13 patients (86%) were in CR/CRu and one (7%) in PR
After a median follow-up of 14 months (range, 5─19), only one patient progressed (not TP53 mutated nor 17p deleted) and 14 patients remained in CR and under treatment.
Measurable residual disease (MRD) was assessed in all evaluable patients:
- At the end of Cycle 3 and at the end of Cycle 6, all the evaluable patients (n = 12, and n = 11, respectively) were MRD negative in peripheral blood
- At the end of Cycle 6 all evaluable patients (n = 10) were MRD negative in the bone marrow
- In comparison with Cohort A and Cohort B, which consisted of R/R MCL patients, the MRD negativity rate was higher in Cohort C (after Cycle 3, 66.5 % vs 78.6% vs 100.0%, and after Cycle 6, 66.5 % vs 77.0% vs 100.0%, respectively). This suggested that the efficacy of the therapy is better if given early in the course of the disease.
The progression-free survival at 1 year was 93.3% (95% confidence interval, 81.5─100.0%) and the overall survival was 100% at 1 and 2 years.
Conclusion
The triplet therapy was well tolerated and effective in untreated patients with MCL, with all evaluable patients remaining MRD negative at the end of Cycle 6 in both blood and bone marrow. These results will be further validated by a larger phase II trial (OAsis II) that is due to start at the end of 2020. This study will compare ibrutinib, venetoclax plus anti-CD20 antibody vs ibrutinib, and anti-CD20 antibody therapy as frontline for adult patients with newly diagnosed MCL.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

