All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
The management of toxicities associated with targeted agents in CLL
The Lymphoma Hub is looking forward to hosting a virtual satellite symposium on the sequencing of therapies in high-risk relapsed/refractory (R/R) lymphoma and chronic lymphocytic leukemia (CLL). The symposium will take place on June 18, 2021, at the 16th International Conference on Malignant Lymphoma (ICML). Lymphoma Hub Chair Gilles Salles will host the symposium, which will include talks from international experts Francesc Bosch, Andrew Davies, Loretta Nastoupil, and Steven Le Gouill.
In the lead-up to the symposium, the Lymphoma Hub will be providing a series of articles to highlight the challenges faced when sequencing therapies for patients with high-risk R/R lymphoma and CLL. This article will focus on the management of toxicities associated with targeted therapies used to treat R/R CLL.
Introduction
Over the past decade, the treatment landscape for CLL has evolved dramatically. Targeted agents such as venetoclax, Bruton’s tyrosine kinase inhibitors (BTKi), and phosphoinositide 3-kinase inhibitors (PI3Ki) have improved response rates among patients with high-risk R/R CLL. However, navigating the side effect and toxicity profiles of these agents has been challenging. An educational session presented by Nicole Lamanno, Kirsten Fischer, and Danielle Brander addressing the management of toxicities of targeted agents was held during the 62nd American Society of Hematology (ASH) Annual Meeting and Exposition.1 The Lymphoma Hub is happy to provide a summary, consolidated with the corresponding literature.
Venetoclax
The Bcl-2 inhibitor venetoclax is approved for the treatment of patients with CLL, both alone and as part of combination regimens. Although venetoclax has demonstrated remarkable survival rates among patients with CLL, there have been a number of toxicities associated with its use (Figure 1).2
Tumor lysis syndrome
Firstly, real world data for venetoclax has brought to light the increased incidence of tumor lysis syndrome (TLS) outside of clinical trials (1–4% in clinical trials vs 6–13% outside of clinical trials). This increase in TLS has been attributed to ‘non-adherence to mitigation measures’, as well as with a number of predisposing factors, including reduced renal function, high tumor burden, and concomitant medications.1
Risk assessment and close monitoring is advised in patients receiving venetoclax-based regimens to reduce the likelihood of TLS. Subsequent effective management of patients involves laboratory monitoring, the use of uricosuric agents, debulking strategies, and dose adjustments. Reducing the bulk of CLL prior to treatment with venetoclax has been shown to decrease the overall tumor burden and consequently improve individual patient risk classifications.1
Neutropenia
Grade 3–4 neutropenia is observed in up to 40%, 60%, and 70% of patients who receive venetoclax as a single agent, in combination with anti-CD20 antibodies, and in combination with BTKi, respectively. These figures highlight the substantial threat neutropenia poses on the use of venetoclax in patients with CLL. Incidences of febrile neutropenia, however, are lower at around 3–5%. The main management strategies for venetoclax-induced neutropenia include pausing treatment, dose reductions, and the use of granulocyte colony-stimulating factor (GCSF).1,2
Other toxicities
Serious infections and gastrointestinal side effects have also been observed in patients treated with venetoclax, but management strategies are less well established. Both toxicities should be addressed on an individual basis.2
Figure 1. Management approaches for venetoclax-associated toxicities*
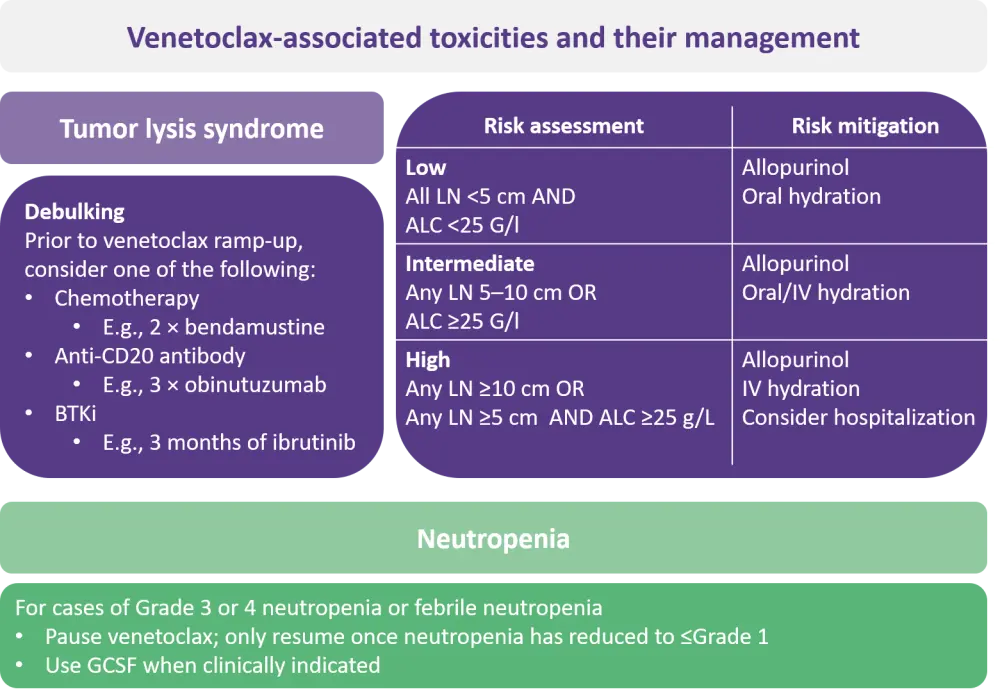
ALC, absolute lymphocyte count; BTKi, Bruton’s tyrosine kinase inhibitor; GCSF, granulocyte colony-stimulating factor; IV, intravenous; LN, lymph node.
*Adapted from Fischer, et al.2
BTK inhibitors
The Lymphoma Hub previously provided a summary of the value of BTKi for the treatment of patients with R/R CLL, where you can find the status of different BTKi in the R/R CLL setting. Here, we focus on the management of toxicities linked to treatment with BTKi.
Like venetoclax, higher incidences of adverse events (AEs) have been observed with the use of BTKi in the real-world setting. Experience in both the United States and Denmark has shown treatment discontinuation reaching 42%, for which AEs were responsible for at least half and were most prevalent in the first 6 months of therapy.1
AEs associated with BTKi can be explained in part by their kinase-binding characteristics. Although the two currently approved agents bind irreversibly to BTK, the design of acalabrutinib advanced that of ibrutinib, displaying higher selectivity to BTK and sparing alternative kinases with a similarly conserved cysteine residue.3 There are a number of third generation, reversible, noncovalent BTKi in early stages of development. Non-covalent binding promises to improve BTKi-associated toxicities and overcome the resistance to BTK inhibition that has been attributed to mutations in the C481 binding site of BTK.2
Ibrutinib has been the most extensively evaluated and has a well-defined toxicity profile. Clinically meaningful long-term AEs associated with ibrutinib include cardiac arrhythmias, bleeding, infection, diarrhea, arthralgias, and hypertension.3 The major toxicities linked to BTK inhibition, and how to manage them, are summarized in Figure 2.
Figure 2. Management of the toxicities associated with BTK inhibitors*
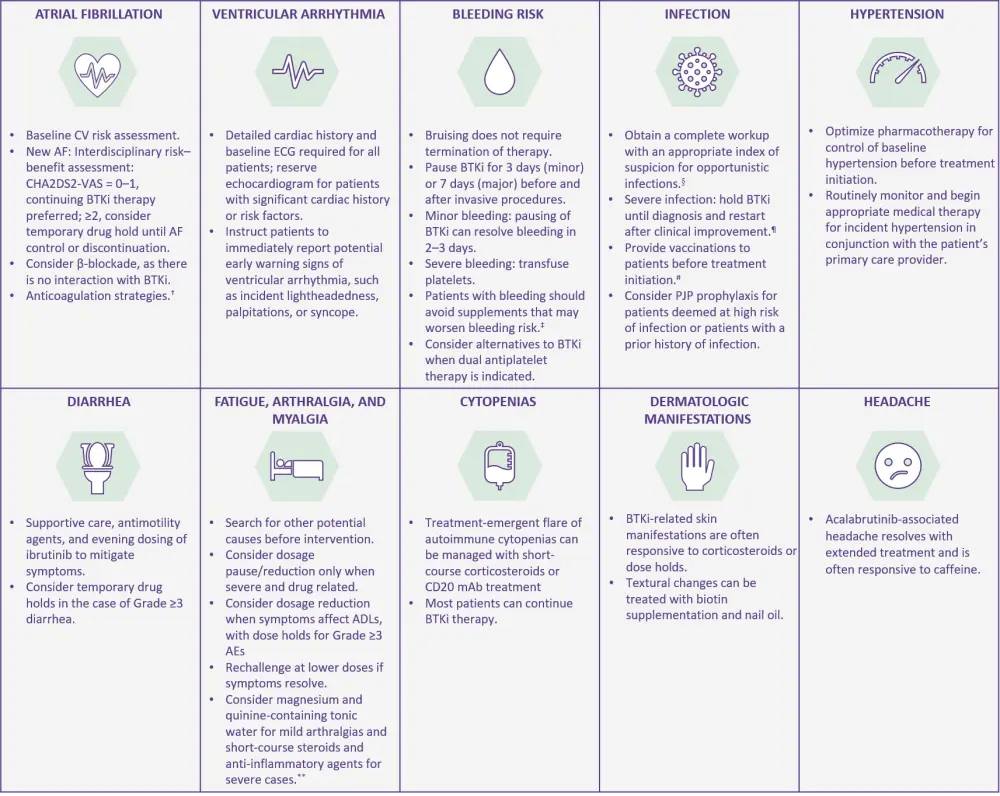
ADLs, activities of daily living; AE, adverse event; AF, atrial fibrillation; BTKi, Bruton’s tyrosine kinase inhibitor; CV, cardiovascular; ECG, electrocardiogram; mAb, monoclonal antibody; PJP, Pneumocystis jirovecii pneumonia.
*Adapted from Lipsky, Lamanna.3
†Avoid combination with vitamin K antagonists.
‡Such as vitamin E or fish oil.
§Such as Aspergillus fumigatus and PJP.
¶Except in cases of fungal infections.
#E.g., against influenza and pneumococcus.
**Evidence anecdotal.
How should we manage toxicities associated with BTK inhibitors in CLL?
PI3K inhibitors4
PI3Ki have demonstrated impressive efficacy in patients with R/R CLL across a number of phase III clinical trials, resulting in the approval of two agents, idelalisib and duvelisib, to date. However, as a result of the unfavorable toxicity profile of PI3Ki, their clinical use has been limited.3
Toxicities associated with PI3Ki can be generally categorized into early- and late-onset complications, as shown in Figure 3.
Figure 3. Toxicities associated with PI3K inhibitors*
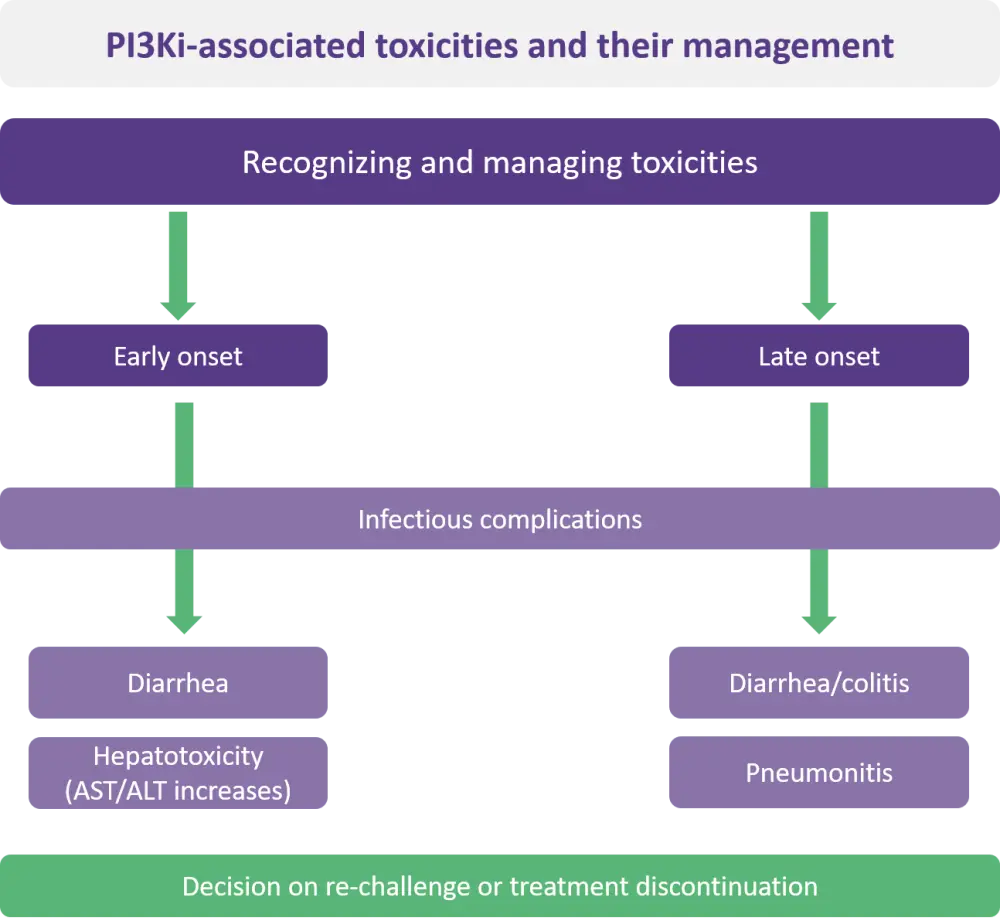
ALT, alanine transaminase; AST aspartate transaminase; PI3Ki, phosphoinositide 3-kinase inhibitor.
*Adapted from Hanlon, Brander.4
Infectious complications and neutropenia
Serious infections that can be fatal are a concern in patients treated with PI3Ki, particularly those who have not received prophylaxis.4 Pneumocystis jirovecii pneumonia (PJP) and cytomegalovirus (CMV) infections are commonly observed and should be treated accordingly. Preferred treatment for PJP is sulfamethoxazole/trimethoprim for 2–6 months following the end of PI3Ki therapy. Patients who develop cytopenias throughout their treatment with a PI3Ki may be offered another PJP prophylaxis regimen due to the myelosuppressive nature of conventional treatments. Prior to, and monthly throughout the course of treatment with a PI3Ki, patients should be monitored for CMV. If detected in significant levels, acyclovir or an equivalent viral prophylaxis can be used. Other viruses should also be assessed prior to treatment with a PI3Ki, including HIV, hepatitis B virus, and hepatitis C virus, for which expert opinion is required.4
Despite incidence rates of neutropenia reaching up to 50% (>20 % Grade 3 or 4) in patients treated with a PI3Ki, treatment cessation or modification as a result is rare.1 Neutropenia should be monitored by a complete blood count every 2 weeks from onset of treatment throughout the first 3 months and should increase to weekly should Grade ≥3 neutropenia develop.4
Hepatotoxicity4
Increased levels of aspartate transaminase or alanine transaminase are indicators of hepatoxicity in patients treated with PI3Ki and are most commonly observed within the first 12 weeks of treatment. Therefore, hepatic monitoring is crucial throughout these early stages of treatment with a PI3Ki for optimal management and outcomes.
Pneumonitis4
Noninfectious pneumonitis can give rise to opportunistic infections in patients treated with PI3Ki, which should be managed using antimicrobial treatments. In the case of severe pneumonitis, PI3K inhibition may be paused and patients may be treated with high-dose corticosteroids.
Conclusion
Impressive efficacy of novel targeted agents has revolutionized the treatment of patients with CLL. Effective management of the toxicities associated with these agents is of utmost importance to improve the survival rates of patients with CLL while maintaining a good quality of life.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

