All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
ASH 2016 | Extension of Phase III RESONATE-2 trial: Single-agent ibrutinib has substantial efficacy in previously untreated elderly CLL/SLL patients
On 3–6 December 2016 in San Diego, CA, the 58th Annual Meeting & Exposition of the American Society of Hematology (ASH) took place. On Saturday 3rd December, an oral abstract session was held between 4:00pm and 5:30pm in the “CLL: Therapy, Excluding Transplantation Program” category. This session was moderated by Tait D. Shanafelt, MD, of the Mayo Clinic, and John F. Seymour, MB BS PhD FRACP, of the Peter MacCallum Cancer Centre and Royal Melbourne Hospital.
Abstract #234 was presented during this session, titled “Updated efficacy and safety from the Phase III Resonate-2 Study: Ibrutinib as First-line Treatment Option in Patients 65 Years and Older with Chronic Lymphocytic Leukemia/Small Lymphocytic Leukemia” by Paul Barr, MD, of the University of Rochester, Wilmot Cancer Institute, Rochester, NY, and colleagues.
The results presented during this talk concerned an extension study, providing updates safety and efficacy results of ibrutinib in the RESONATE-2 trial; a randomized, Phase III trial with the aim of comparing efficacy and safety of ibrutinib and chlorambucil in previously untreated CLL/SLL patients.
Eligible patients with previously untreated CLL/SLL, aged ≥65 years, were randomized 1:1 to receive 420mg ibrutinib daily until progression or 0.5mg/kg chlorambucil (max 0.8mg/kg) on Days 1 and 15 of a 28-day cycle up to 12 cycles. PFS per iwCLL 2008 criteria, with 2012 clarification for treatment related lymphocytosis, was the primary endpoint. All patients in the PCYC-1115 study rolled over to the PCYC-1116 extension study and chlorambucil arm patients who experienced disease progression could cross-over to ibrutinib or investigator’s choice. Two-hundred and sixty-nine patients were included, with a median age of 73 years (70% were ≥70 years of age); baseline characteristics were balanced between the ibrutinib and chlorambucil arms.
Highlights:
- Median follow-up = 28.6 months
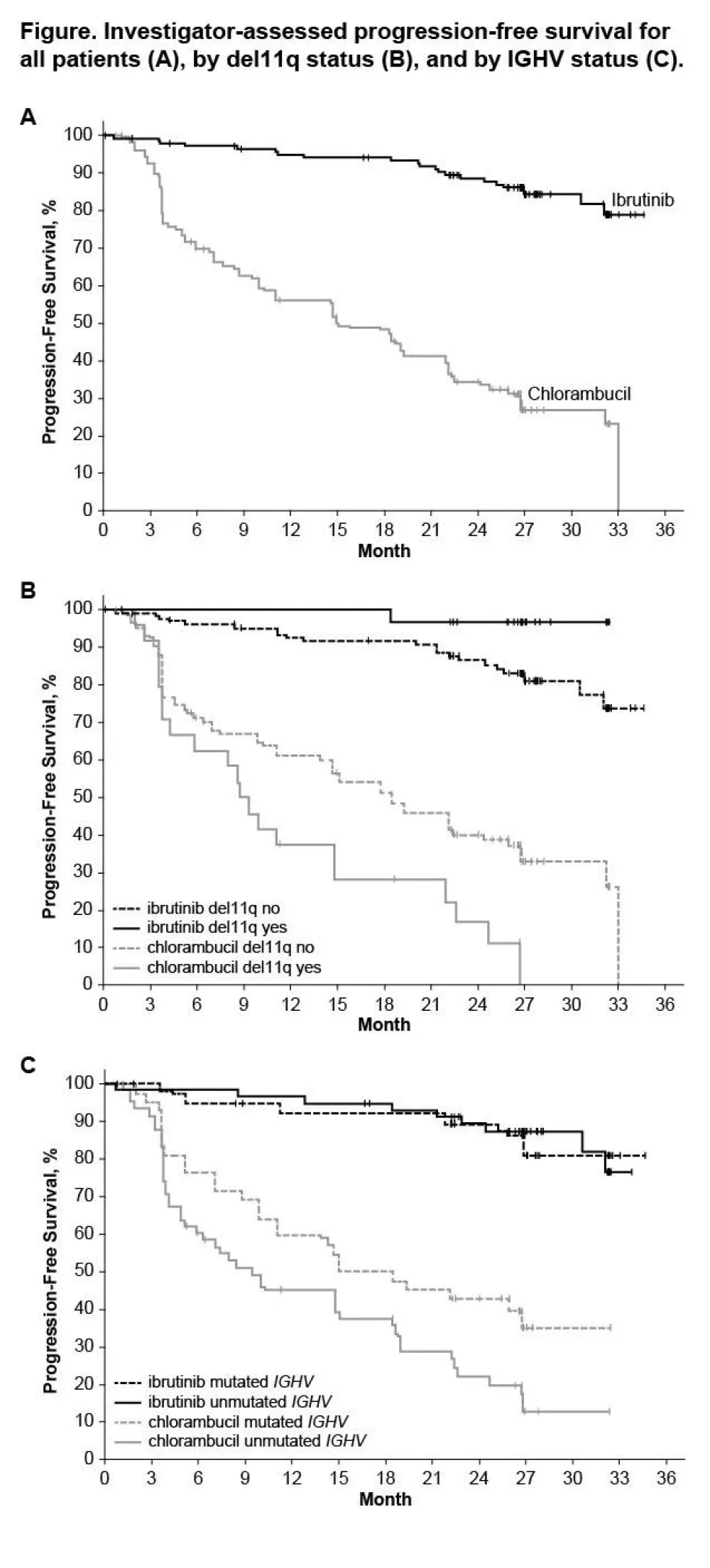
- Prolongation of PFS for ibrutinib versus chlorambucil was sustained (89% vs 34% at 24-months; HR 0.121; 95% CI 0.074–0.198; P<0.0001)
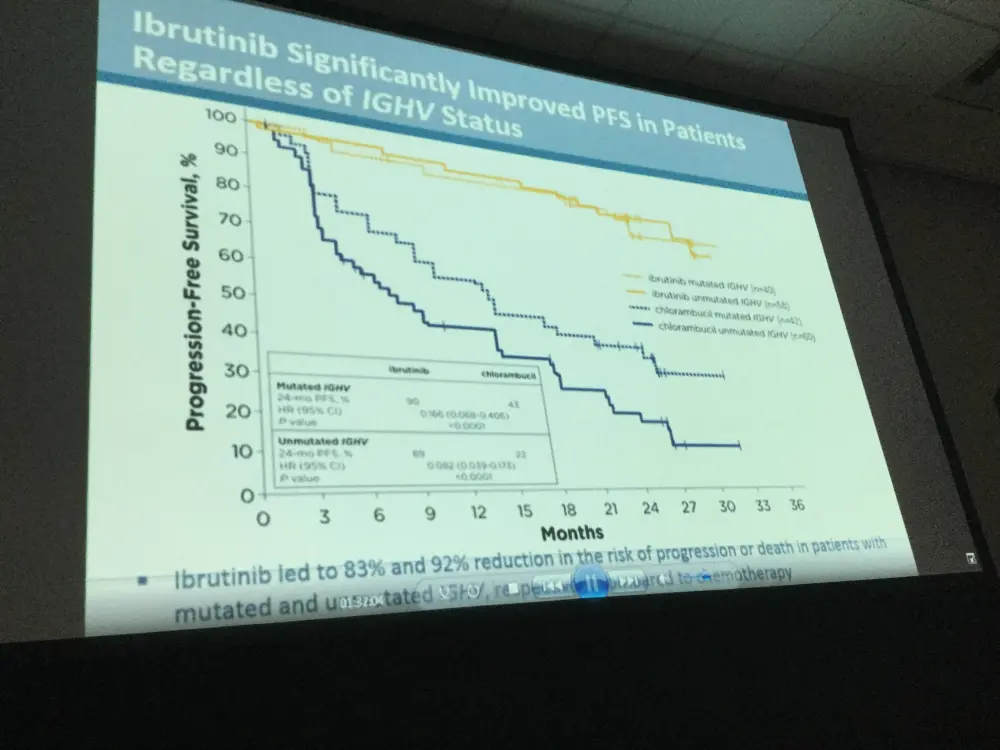
- PFS was significantly higher in high-risk subgroups, including del11q and unmutated IGHV gene, when treated with ibrutinib compared to chlorambucil
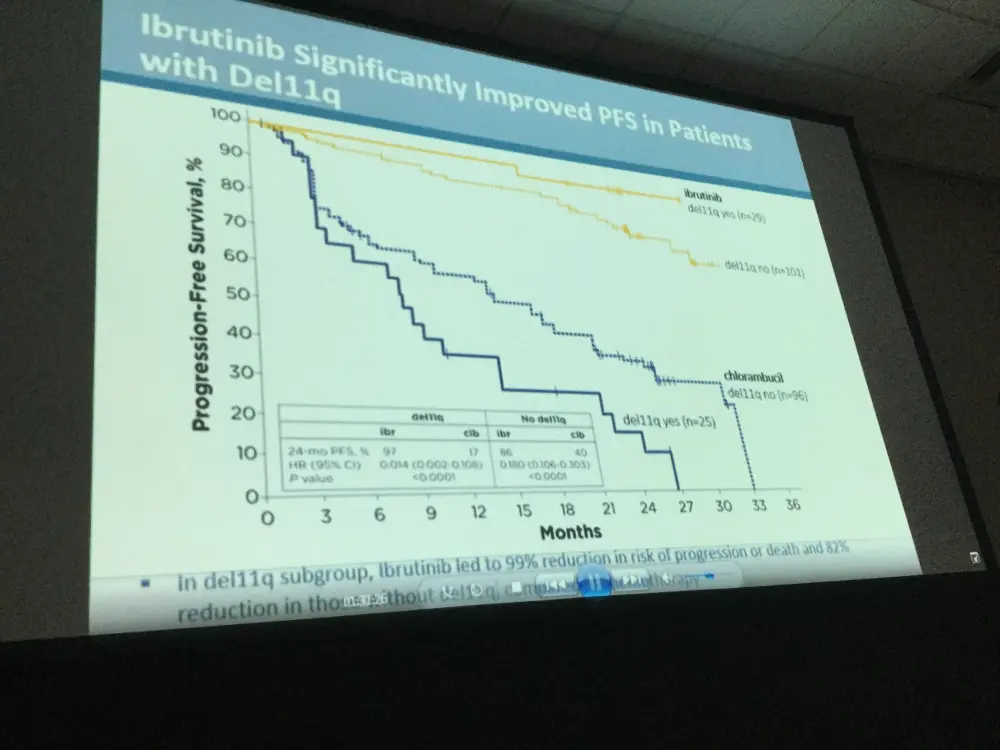
- Due to disease progression, 4/135 patients discontinued treatment, 1 case of Richter’s transformation was reported in each treatment arm
- Forty-one percent of patients crossed-over from chlorambucil to ibrutinib
- OS analysis in ITT population resulted in 2-year survival rate estimates of 95% for ibrutinib and 84% for chlorambucil arms, respectively
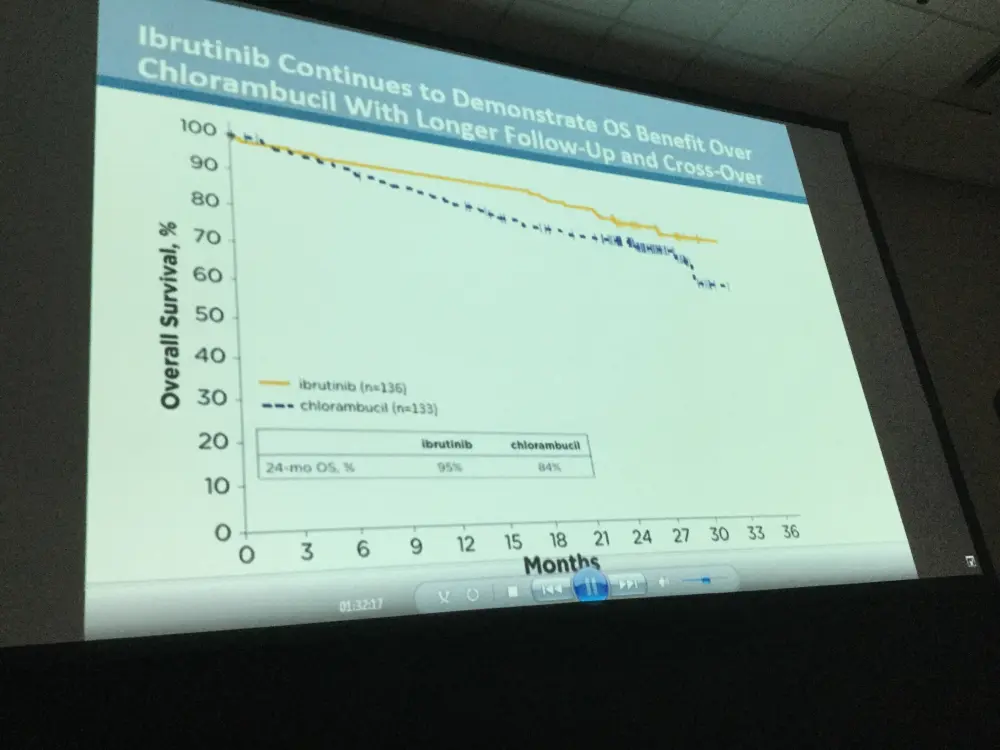
- Investigator-assessed ORR for ibrutinib compared to chlorambucil was 92% versus 36% (P<0.0001)
- CR/CRi in patients treated with ibrutinib increased from 11% at 18.4 months to 18% with longer follow-up of 28.6 months
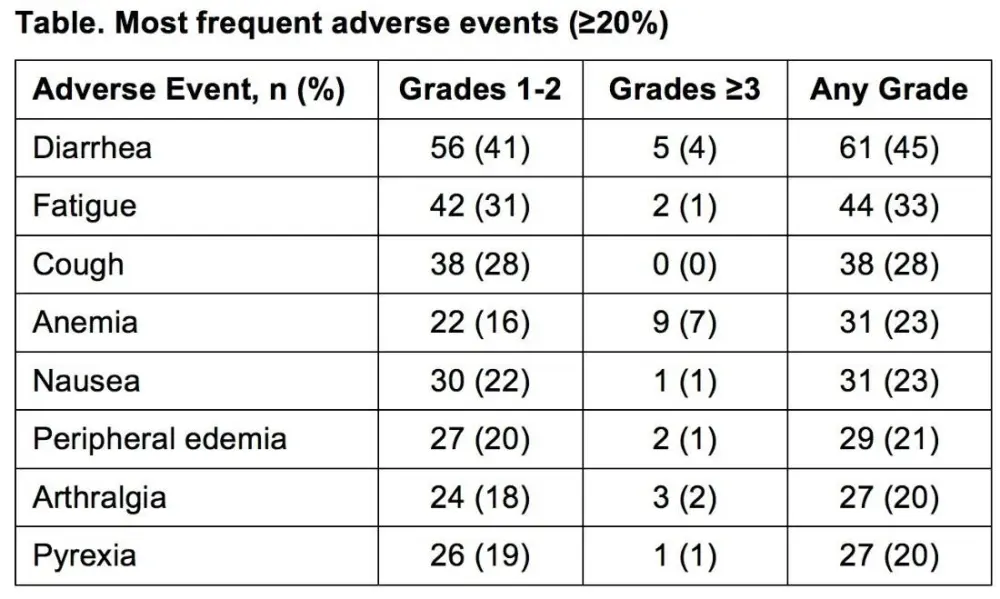
- In the ibrutinib arm, ≥ grade 3 AEs in ≥ 5% patients included: neutropenia (12%), pneumonia (7%), anemia (7%), and hypertension (5%)
- Dose reductions due to AEs (of any grade) took place in 13% patients
- AEs were the most common reason for treatment discontinuation (12%); most occurred in the first 12-months of treatment with ibrutinib

This abstract concluded by stating that with a longer follow-up, ibrutinib continued to have substantial efficacy, reducing risk of progression or death by 88%. Quality of responses also improved over time; 18% of patients achieving a CR/CRi on ibrutinib. Finally, even with a high rate of patients switching from chlorambucil to ibrutinib therapy, OS remained significantly improved for patients who were randomized to receive ibrutinib.
Abstract
Background: Chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL) occurs primarily in older patients (pts) who often have increased comorbidities and cannot tolerate aggressive treatments, which leads to poorer outcomes (Balducci, Cancer Control 2015; Thurmes, Leuk Lymphoma 2008). Alkylating agents, such as chlorambucil (clb), have been commonly used to treat these pts (Eichhorst, Blood 2009). Ibrutinib (ibr), a first-in-class, once-daily, inhibitor of Bruton’s tyrosine kinase, is indicated by the US FDA for the treatment of pts with CLL/SLL and allows for treatment without chemotherapy. RESONATE-2 (PCYC-1115) is a randomized phase 3 trial designed to compare the efficacy and safety of ibr vs clb in pts with treatment-naïve (TN) CLL/SLL (Tedeschi, ASH 2015). Primary results, as assessed by an independent review committee (IRC), demonstrated with a median follow-up of 18.4 mo that ibr significantly reduced the risk of progression or death by 84% (P<0.001) (Burger, N Engl J Med 2015). Based on these results, ibr was approved as a first-line treatment option for CLL pts. This extension study provides updated efficacy and safety results of ibr for RESONATE-2.
Methods: Eligible pts with TN CLL/SLL aged ≥65 y were randomized 1:1 to receive 420 mg ibr daily until progression or 0.5 mg/kg clb (max 0.8 mg/kg) on days 1 and 15 of a 28-d cycle for up to 12 cycles. Pts with del17p were excluded. Pts were stratified by ECOG status and Rai stage. The primary endpoint was PFS per iwCLL 2008 criteria, with 2012 clarification for treatment related lymphocytosis. Secondary endpoints included OS, ORR, rate of hematologic improvement, and safety; longer term follow-up safety data focused on ibr. Pts in the 1115 study with progressive disease (PD) were enrolled in the PCYC-1116 extension study. At 1115 study closure, all pts in 1115 rolled over to 1116. Pts on the clb arm with PD could cross-over to ibr or investigator’s choice.
Results: Median age of the 269 pts was 73 y (70% ≥70 y). Baseline characteristics were balanced between arms; 45% had advanced Rai stage, 20% had del11q, and 69% had comorbidities at baseline, including CIRS score >6, reduced creatinine clearance, or ECOG performance status of 2. With a median follow-up of 28.6 mo, prolongation of PFS for ibr vs clb was sustained (89% vs 34% at 24-mo; HR, 0.121; 95% CI 0.074-0.198; P<0.0001; Figure). PFS was significantly improved for ibr across high-risk subgroups, including del11q and unmutated IGHV gene (Figure). Overall, 4 of 135 pts discontinued ibr due to PD. 1 case of Richter’s transformation was observed in each arm. With 41% of pts switching from clb to ibr, the OS analysis in the ITT population resulted in 2-yr survival rate estimates of 95% and 84% in the ibr and chlorambucil arms, respectively. The investigator-assessed ORR was 92% with ibr vs 36% with clb (P<0.0001). CR/CRi within the ibr arm improved from 11% at 18.4 mo to 18% with longer follow-up of 28.6-mo. Sustained hematological improvements were higher for ibr vs clb for those with anemia (90% vs 45%; P<0.0001) or thrombocytopenia (80% vs 46%; P=0.0055) at baseline. The most frequent adverse events in ibrutinib treated pts (AEs; ≥20%) are presented in the Table. Grade [Gr] ≥3 AEs in ≥5% of pts included neutropenia (12%), pneumonia (7%), anemia (7%), and hypertension (5%); AEs (any Gr) leading to dose reductions occurred in 13%. The most common reason for discontinuation was AEs (12%), with most occurring during the first yr of ibr therapy. The incidence of the common Gr ≥3 AEs in these ibr-treated pts typically decreased over time. Major hemorrhage occurred in 7% (1 Gr 2, 7 Gr 3, 1 Gr 4; 5 in first 12 mo and 4 between 1-2 y) and atrial fibrillation occurred in 10% (1 Gr 1, 7 Gr 2, 6 Gr 3) of ibr-treated pts. With a median treatment duration of 28.5 mo (range, 1-36), 79% of pts remain on first-line ibr.
Conclusions: With a median time on study of 28.6 mo, ibr continued to have substantial efficacy, with 88% reduction in risk of progression or death. Furthermore, the quality of responses has improved over time, with 18% of CLL/SLL pts achieving a CR/CRi with single agent ibr. Treatment limiting AEs decreased in frequency with longer follow-up, with 79% of this elderly pt population continues daily ibr. Lastly, even with a high rate of cross-over in the clb arm, OS remains significantly improved for pts randomized to ibr.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

