All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Incyte, Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Challenges in R/R CLL and DLBCL: Overcoming drug resistance
The Lymphoma Hub is looking forward to hosting a virtual Satellite Symposium on the management and treatment of patients with relapsed/refractory (R/R) lymphoma. The symposium will take place on Sunday, November 8, 2020, at the European School of Haematology (ESH) conference, How to Diagnose and Treat Lymphoma, where a panel of international experts, Marie José Kersten, Kieron Dunleavy, Francesc Bosch, Astrid Pavlovsky, and the Lymphoma Hub Chair, Gilles Salles, will provide insight into their experiences managing patients with R/R disease. This article will address the issue of drug resistance in the treatment of diffuse large B-cell lymphoma (DLBCL) and chronic lymphocytic leukemia (CLL).
Figure 1. Overview of the challenges and treatment approaches when managing patients with R/R lymphoma
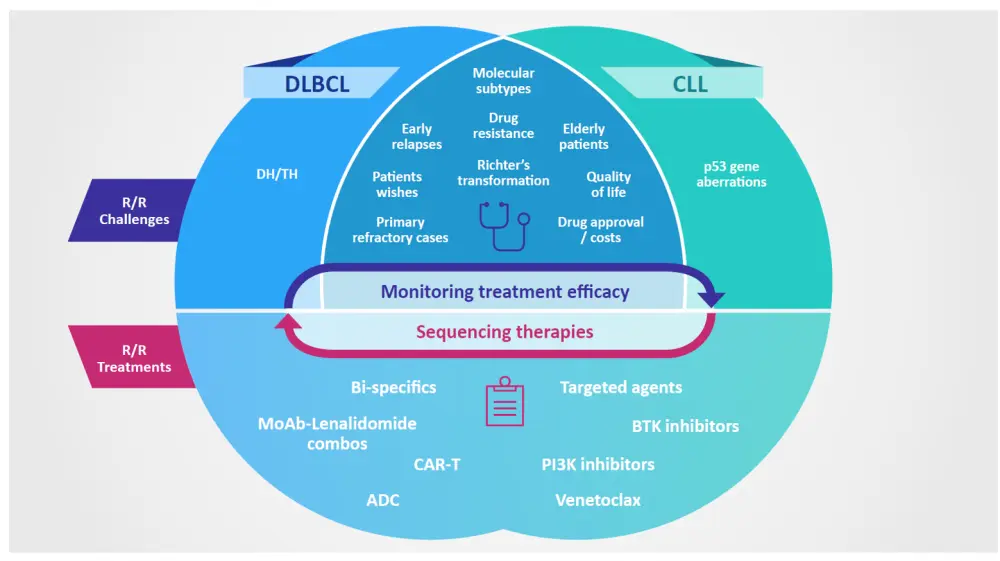
ADC, antibody-drug conjugate; BCR, B-cell receptor; BTK, Bruton’s tyrosine kinase; CAR, chimeric antigen receptor; CLL, chronic lymphocytic leukemia; DH/TH, double hit/triple hit; DLBCL, diffuse large B-cell lymphoma; PI3K, phosphoinositide 3-kinase.
Schematic courtesy of Gilles Salles.
Challenges and treatment approaches when managing patients with R/R lymphoma
If you would like to download this resource, click below.
Despite recent advances in treatments for lymphoma and the resulting positive impact on patient prognosis, a proportion of patients develop resistance to their anti-lymphoma therapy. Research into the disease biology has resulted in increased understanding of the mechanisms behind drug resistance and subsequently the design of novel agents to tackle it. Here, we discuss patient resistance to some of the most widely utilized therapies in lymphoma, and the problems surrounding it.
DLBCL
R-CHOP
Rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) remains the most frequently employed frontline treatment for DLBCL; however, between 20–50% of patients develop chemoresistance or relapse.1 Data from a large-scale differential multi-omics study uncovered 22 transcripts whose expression levels differed significantly between patients who develop R/R disease and those who did not. The study identified regulators of tumor metabolism (Hexokinase 3), microenvironment (IDO1, CXCL13), proliferation, migration and invasion (S100 proteins), and B-cell receptor (BCR) signaling pathway (CD79B) as particularly prominent biomarkers for chemoresistance; thus, uncovering several potential therapeutic targets.2 Although research remains in early stages, understanding the factors that influence drug resistance in the frontline setting may help to minimize the number of patients who develop R/R disease, and therefore improve prognosis globally within the DLBCL setting.
Rituximab
Since its approval almost 20 years ago, the anti-CD20 monoclonal antibody, rituximab, remains the only agent to improve upon the cyclophosphamide, doxorubicin, vincristine, and prednisolone (CHOP) regimen for the treatment of DLBCL.3 Application of rituximab also extends into the treatment of R/R disease and has transformed the treatment landscape. However, resistance to rituximab is a common complication, and so researchers have designed a number of next generation CD20-directed antibody-based therapies hoping to overcome this. Even so, no novel agents have replaced rituximab as either a component of the R-CHOP frontline regimen nor a leading treatment for R/R disease.3,4
The scope for CD20-directed therapy is not limited to antibody-based approaches, and a novel engineered toxin body, MT-3724, has demonstrated promising efficacy in a phase II clinical study (NCT02361346). MT-3724 consists of a CD20-directed single chain variable fragment and a Shiga-like toxin A. This innovative combination provides high selectivity and enables internalization and cell death via inactivation of ribosomal protein synthesis. Therefore, MT-3724 aims to overcome the non-internalizing nature of CD20 which has contributed to drug resistance. The agent has shown promising on-target effects in vivo and clinical efficacy in patients with non-Hodgkin lymphoma (NHL).5 Could MT-3724 be a viable option for patients with R/R DLBCL?
CLL6
In CLL, agents targeting BCR signaling, Bruton's tyrosine kinase (BTK), and BCL-2, are at the forefront of R/R disease.
Ibrutinib
The BTK inhibitor ibrutinib has demonstrated clinical efficacy in both patients with R/R CLL and those with high-risk untreated disease. Of the patients who achieve a good response to ibrutinib initiation, 8–13% acquire secondary resistance, after which patient prognosis is poor. Mechanisms of resistance to ibrutinib have been widely investigated, and a number of mutations have been associated with post-ibrutinib CLL relapse (summarized in Figure 2). Owing thanks to the increased understanding of disease biology, novel therapeutic interventions can minimize the risk of ibrutinib resistance (Figure 2). Detection of BTK, PLCG2, and BCL2 mutations could be a useful prognostic marker for predicting relapse to ibrutinib, as they occur several months prior to relapse.
Epigenetic modulation represents one of the prospective approaches to overcoming resistance to ibrutinib, and the Lymphoma Hub presented a summary of how histone deacetylase inhibitors could reduce BTK expression and alleviate ibrutinib resistance in patients with CLL, read it here.
Figure 2. Mechanisms of resistance to ibrutinib, and current and prospective treatment approaches for patients who develop resistance to ibrutinib
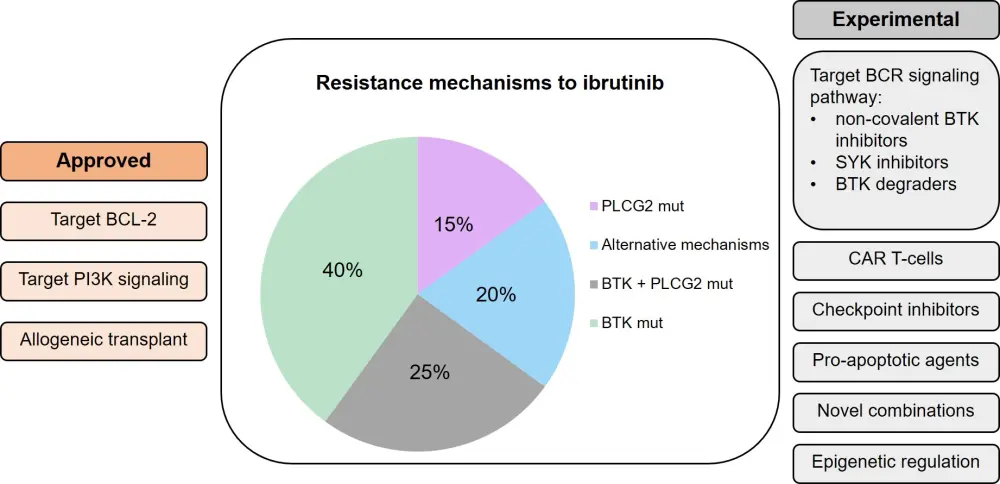
BCR, B-cell receptor; BTK, Bruton’s tyrosine kinase; CAR, chimeric antigen receptor; PI3K, phosphoinositide 3-kinase; SYK, spleen tyrosine kinase.
Adapted from Sedlarikova et al.6
Venetoclax
The oral BCL-2 antagonist, venetoclax, is now a widely used therapy for patients with CLL with TP53 disruption or those who have relapsed following ibrutinib and/or immunochemotherapy. However, there are also a number of known mechanisms of resistance to venetoclax, including mutations in BCL-2 itself, and in genes encoding other signaling proteins such as TP53, NOTCH1, and BTG1. One way to prevent development of venetoclax resistance in CLL is identifying clinically effective combination regimens which are administered for a limited time. In fact, the combination of ibrutinib with venetoclax appears promising in the R/R CLL setting. It has been shown that CLL cells previously treated with BCR inhibitors elicit an increased dependence on BCL-2, and venetoclax appears to augment BTK inhibitor-mediated CLL cell apoptosis. The precise mechanisms of synergy remain unclear and are under investigation.
Conclusion
Drug resistance remains a problem for patients with DLBCL. A variety of factors have been associated with an increased risk of developing resistance, providing rationale for a number of novel therapeutic targets. It is important that, in addition to establishing novel treatments, the techniques used to identify resistance-driving mutations are refined. Early identification of such mutations can guide treatment decisions, resulting in superior patient prognoses.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

