All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Fixed duration or MRD-guided venetoclax maintenance treatment in patients with CLL unfit for first-line FCR: Results from HOVON 139/GIVE trial
High rates of undetectable minimal residual disease (uMRD) have been documented in fixed-duration treatment with venetoclax combined with anti-CD20 antibody in patients with chronic lymphocytic leukemia (CLL) in both the first-line induction therapy (12 cycles) and second-line or relapsed/refractory therapy (24 cycles) settings. The National Institute for Health and Care Excellence (NICE) has recommended venetoclax in combination with obinutuzumab as first-line treatment for patients with previously untreated CLL or patients with CLL without del(17p)/TP53 mutation who are ineligible for treatment with fludarabine, cyclophosphamide, and rituximab (FCR); however, the benefit of maintenance therapy with fixed duration venetoclax after venetoclax + obinutuzumab is not known. During the European Hematology Association (EHA)2021 Virtual Congress, Mark-David Levin1 presented the primary analysis of the HOVON 139/GIVE trial (NL5871) investigating the efficacy of fixed duration or MRD-guided venetoclax maintenance treatment.
Study design
This was a phase II randomized controlled trial in patients with previously untreated CLL who were considered unfit to receive first-line FCR. All eligible patients received preinduction therapy with obinutuzumab and induction therapy with venetoclax + obinutuzumab (Figure 1). Patients achieving complete remission (CR), or partial remission (PR) were then randomized to the following maintenance treatment:
- Arm A: 12 cycles of venetoclax irrespective of MRD status
- Arm B: MRD-guided venetoclax with treatment limited to patients with detectable MRD after induction
Maintenance with venetoclax was stopped if patients reached blood MRD negativity at any timepoint following randomization; patients who were MRD negative at randomization did not start venetoclax treatment.
MRD was centrally assessed by flow cytometry in bone marrow (BM) after 12 and 24 cycles, and in peripheral blood (PB) at screening, Cycles 6 and 12, and every 3 months until Month 27. The primary endpoint was uMRD status (<1:10.000 leukocytes) assessed by flow cytometry in BM after 12 cycles and no disease progression, according to International Workshop on Chronic Lymphocytic Leukemia criteria, after 24 cycles of venetoclax.
Figure 1. Treatment schema*
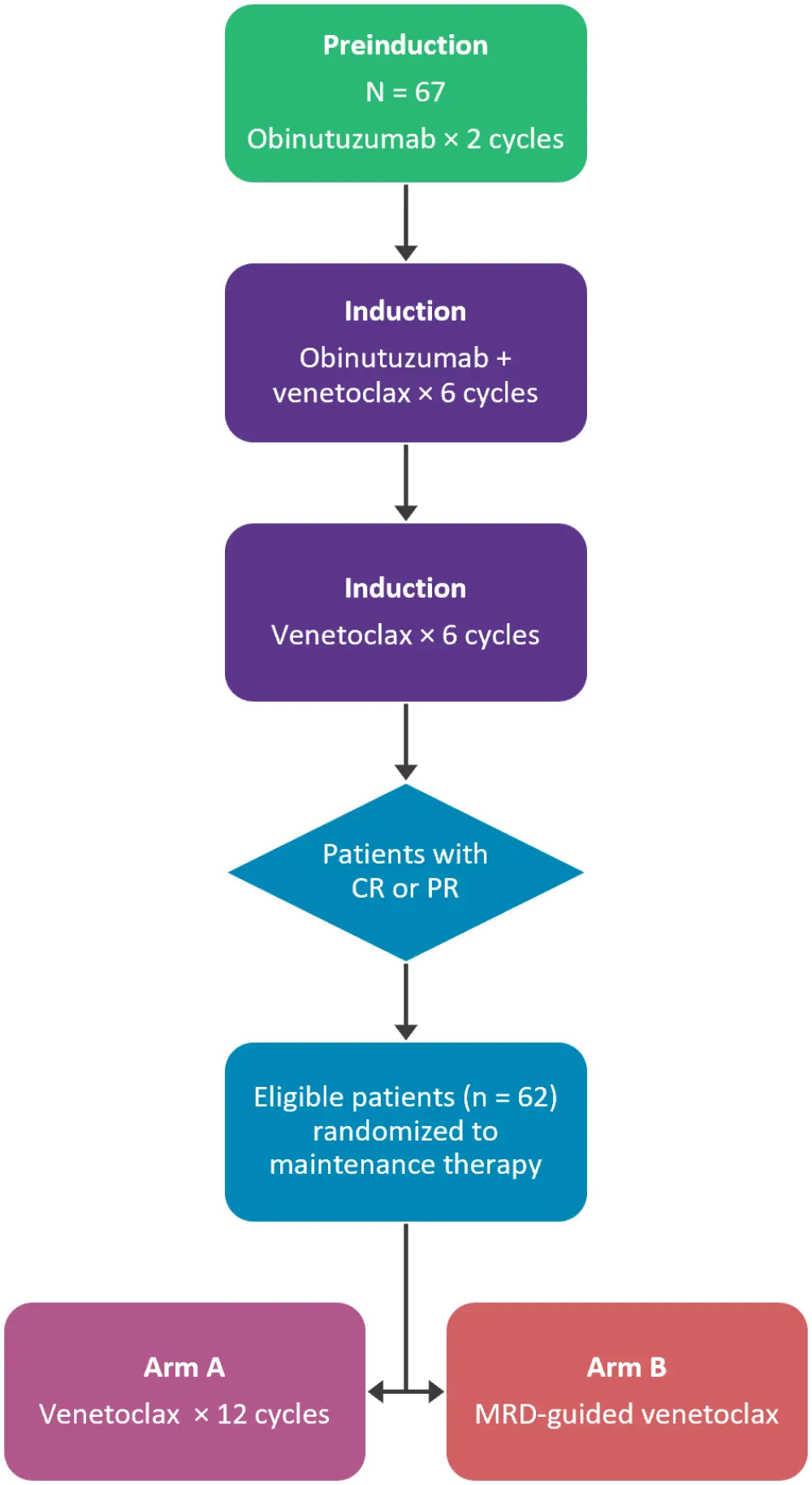
CR, complete remission; MRD, minimal residual disease; PR, partial remission.
*Adapted from Levin, et al.1
Baseline characteristics
There were 67 patients included in the study and the median age was 71 years (range, 57-89 years); 70% of patients were male. The baseline characteristics are presented in Table 1.
Table 1. Baseline characteristics*
|
ALC, absolute lymphocyte count; CIRS, cumulative illness rating scale; CLL-IPI, chronic lymphocytic leukemia international prognostic index; TLS, tumor lysis syndrome. |
|
|
Characteristic, % (unless otherwise stated) |
All patients |
|---|---|
|
Mutational status |
|
|
Mutated |
44 |
|
Unmutated |
56 |
|
TP53 aberration (del(17p) and/or TP53mut) |
13 |
|
CLL-IPI score |
|
|
Low risk (0–1) |
3 |
|
Intermediate risk (2–3) |
17 |
|
High risk (4–6) |
65 |
|
Very high risk (7–10) |
15 |
|
TLS risk |
|
|
Low (lymph nodes <5 cm and ALC <25 × 109/L) |
12 |
|
Intermediate (lymph nodes ≥5 cm and <10 cm or ALC ≥25 × 109/L) |
60 |
|
High (lymph nodes ≥5 cm and <10 cm or >10 cm and ALC ≥25 × 109/L) |
28 |
|
Median CIRS score (range) |
3 (0–16) |
Results
Preinduction treatment
After two cycles of obinutuzumab, tumor lysis syndrome (TLS) risk was reduced from 28% and 60% to 1% and 15% (high and medium TLS risk, respectively).
Induction treatment
Overall response rate (ORR) after induction treatment demonstrated CR in 32% and PR in 67% of patients. At the end of 12 cycles of venetoclax induction therapy, uMRD was detected in 88% and 79% of patients in PB and BM, respectively (Figure 2). There were no notable differences in the following MRD high risk groups in either treatment arm: immunoglobulin heavy chain variable mutated vs immunoglobulin heavy chain variable unmutated (96% vs 93%), del(17p) and/or TP53 mutated vs del(17p) and/or TP53 unmutated (94% vs 100%), and no genomic complexity vs genomic complexity (89% vs 86%).
Figure 1. MRD status after induction therapy*
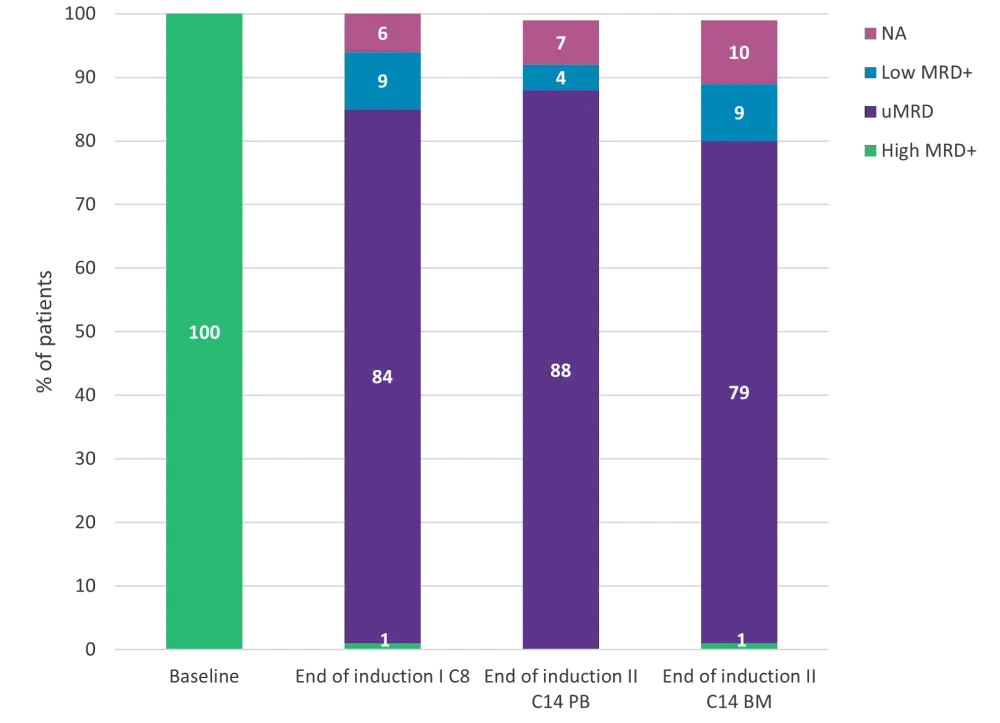
BM, bone marrow; C, cycle; CLL, chronic lymphocytic leukemia; PB, peripheral blood; uMRD, undetectable minimal residual disease.†
*Adapted from Levin, et al.1
†MRD categories (international standardized approach): low (<10−4, i.e., <1 CLL cell per 10,000 leukocytes), intermediate (≥10−4 and <10−2), and high (≥10−2).
Maintenance treatment
A total of 62 patients were eligible for consolidation treatment: 32 patients were randomized to fixed venetoclax treatment and 30 were randomized to MRD-guided venetoclax treatment. In arm A, 28 of the 32 patients received 12 cycles of venetoclax, while only one of the 30 patients in arm B received three cycles of venetoclax, after which time that patient was MRD negative.
- Overall, 59% and 72% of patients in arm A and 56% and 57% of patients in arm B demonstrated uMRD in BM and PB, respectively (Table 2).
- At 27 months, MRD negativity was better in PB compared with BM (72% vs 59%) in arm A.
- An ORR of 88% (16 CR, 12 PR, and 3 progressive disease) was observed in arm A and an ORR of 96% (19 CR, 10 PR, and 1 progressive disease) was observed in arm B.
Table 2. MRD at primary endpoint*
|
BM, bone marrow; MRD, minimal residual disease; PB, peripheral blood; uMRD, undetectable minimal residual disease. |
||
|
MRD, % |
Arm A |
Arm B |
|---|---|---|
|
BM at end of venetoclax Cycle 12 |
|
|
|
uMRD |
84 |
87 |
|
High MRD+ |
0 |
3 |
|
Low MRD+ |
13 |
7 |
|
BM at 27 months |
|
|
|
uMRD |
59 |
57 |
|
High MRD+ |
6 |
3 |
|
Low MRD+ |
13 |
30 |
|
PB at end of venetoclax Cycle 12 |
|
|
|
uMRD |
94 |
97 |
|
High MRD+ |
0 |
0 |
|
Low MRD+ |
6 |
3 |
|
PB at 27 months |
|
|
|
uMRD |
72 |
57 |
|
High MRD+ |
9 |
3 |
|
Low MRD+ |
12 |
33 |
Adverse events
Grade 2 and 3 adverse events were more common in the fixed duration venetoclax arm compared with the MRD-guided venetoclax arm. The most common adverse event was infections (Grade 2, 42% in arm A vs 17% in arm B; Grade 3, 6% vs 3%), particularly pulmonary infections (Grade 2, 13% in arm A vs 0% in arm B; Grade 3, 3% vs 0%).
Conclusion
This phase II trial demonstrated that two cycles of preinduction obinutuzumab reduces TLS risk in most patients prior to treatment with venetoclax + obinutuzumab. The combination of venetoclax + obinutuzumab as induction treatment was highly effective in first-line FCR-unfit patients irrespective of any high-risk features. Both maintenance treatments—fixed duration venetoclax and MRD-guided venetoclax—were well tolerated and demonstrated high efficacy after 24 cycles. MRD-guided venetoclax maintenance treatment also demonstrated low rates of adverse events compared with fixed duration venetoclax. Further research is warranted to investigate the effectiveness of fixed duration and MRD-guided venetoclax maintenance treatment after venetoclax + obinutuzumab induction
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content

