All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Humoral and cellular responses in patients with lymphoid malignancies receiving SARS-Cov-2 vaccination
Factors that influence the efficacy of Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) vaccination in patients with lymphoid malignancies are still not fully detailed. Immune dysregulation associated with active malignancies has proven to impair serological response, and concurrent anti-cancer treatment may also negatively influence humoral immune activity. Previous research has excluded the measurement of T-cell mediated immune responses following vaccination, which could play a protective role against infection irrespective of seropositivity.
Recent papers by Marasco, et al.1 published in BJH, and Liebers, et al.2 in Blood, examined both humoral and T-cell mediated responses in patients with lymphoid malignancies following two doses of SARS-CoV-2 vaccine. We summarize key results from both papers below.
Marasco, et al.1
This was a prospective study observing the efficacy of two doses of either mRNA-1273 or BNT162b2 vaccines administered 28 days apart in adult patients vaccinated at the Istituto Nazionale dei Tumori in Milan, Italy. The general study design is shown below.
Figure 1. Study design
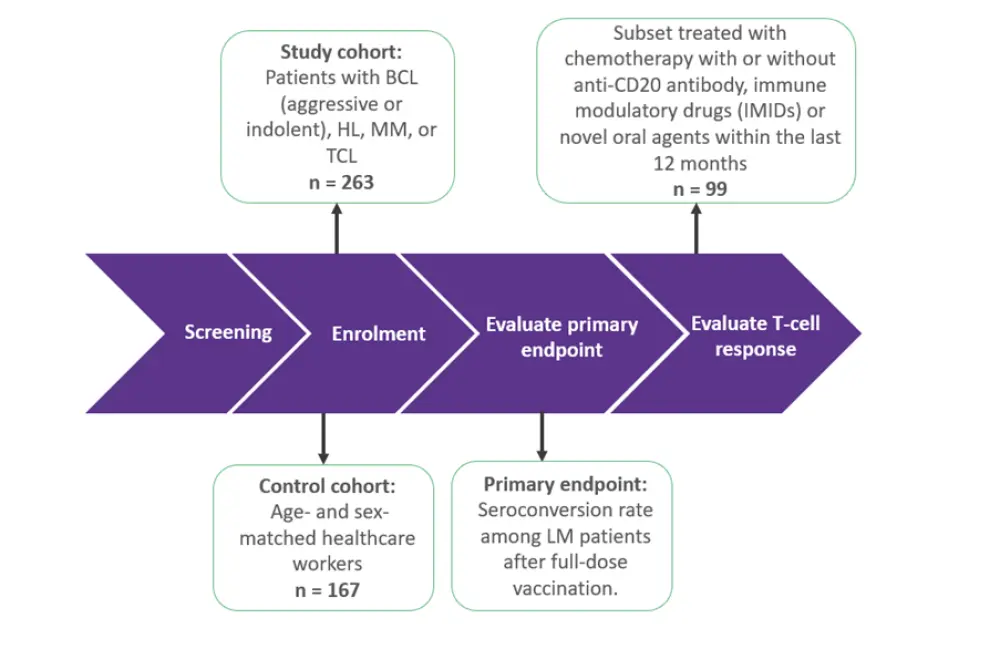
BCL, B-cell lymphoma; HL, Hodgkin lymphoma; IMIDs, immunomodulatory imide drugs; LM, lymphoid malignant; MM, multiple myeloma; TCL, T-cell lymphoma.
*Adapted from Marasco, et al.1
Results
As reported in Table 1, a total of 131 (49.8%) patients achieved seroconversion 4 weeks following first dose while a further 39 patients achieved seroconversion 2 weeks after their second dose (14.8%), producing a total response rate of 64.6%. The median antibody titer at 2 weeks after the second dose was 175 U/ml.
Table 1. Patient characteristics and the associated seroconversion rate 4 weeks after a first vaccine dose and 2 weeks after a second dose*
|
ALC, absolute lymphocyte count; ANC, absolute neutrophil count; CAR-T, chimeric antigen receptor T-cell therapy; HSCT, hematopoietic stem cell transplantation; IgA, immunoglobulin A; IgG, immunoglobulin G; IgM, immunoglobulin M; IMIDs, immunomodulatory imide drugs. |
|||
|
Characteristic, % |
N = 263 |
Seroconversion at 4 weeks |
Seroconversion at 6 weeks |
|---|---|---|---|
|
Overall population |
100 |
49.8 |
64.6 |
|
Female |
46.7 |
49.6 |
63.4 |
|
Diagnosis |
|||
|
B-cell aggressive lymphomas |
22.4 |
23.7 |
35.5 |
|
B-cell indolent lymphomas |
42.4 |
44.1 |
61.2 |
|
Hodgkin lymphoma |
12.6 |
54.5 |
78.8 |
|
Multiple myeloma |
19.8 |
88.4 |
94.2 |
|
T-cell lymphomas |
3 |
50 |
75 |
|
Active disease |
68 |
51.9 |
66.4 |
|
Last therapy ≤12 months prior |
64.2 |
39.6 |
55 |
|
<6 months prior to first vaccine |
53.2 |
37.1 |
53.6 |
|
>6 to ≤12 months prior to first vaccine |
11 |
51.7 |
62 |
|
Last type of therapy |
|||
|
Patients receiving anti CD20 antibody + chemotherapy |
19.3 |
5.9 |
17.6 |
|
Chemotherapy alone |
13.6 |
47.2 |
69.4 |
|
Received IMIDs |
9.9 |
80.7 |
84.6 |
|
Novel oral agents |
8 |
23.8 |
52.3 |
|
Receiving CAR-T cell therapy or HSCT |
8 |
52.3 |
57.1 |
|
ALC, <800 cells/µl |
18.6 |
22.4 |
42.8 |
|
ANC, <1500 cells/µl |
7.6 |
35 |
60 |
|
IgG, <600 mg/dl |
21.3 |
42.8 |
53.5 |
|
IgA, <80mg/dl |
28.5 |
44 |
57.3 |
|
IgM, <40mg/dl |
32.3 |
40 |
51.7 |
Patients receiving anti-CD20 treatment plus chemotherapy had the lowest seroconversion rate and median antibody titers 2 weeks after the second vaccine dose. This was significantly lower than patients who received other treatments (17.6% vs 71.2%; odds ratio [OR], 0.09; 95% confidence interval [CI], 0.04–0.20; p < 0.001; median antibody titre, 0.44 U/ml vs 183 U/ml; p < 0.0001). Patients with multiple myeloma treated with immunomodulatory imide drugs, in contrast, had a higher seroconversion rate.
Another notable observation was the effect of the time interval between last treatment and first vaccine dose. Patients who received their last treatment within 6 months of vaccination had comparable seroconversion to those who received their last treatment >6 months to ≤12 months, suggesting long lasting immunosuppression.
A comparison of 167 patients with lymphoid malignancies with 167 healthy controls revealed significantly lower seroconversion (Figure 2) among the patients with lymphoid malignancies, as well as a lower antibody titer two weeks following a second dose of the COVID-19 vaccine (median, 1,078 U/ml vs 207.5 U/ml; p < 0.001).
Figure 2. Comparison of seroconversion between patients with lymphoid malignancies and healthy controls*
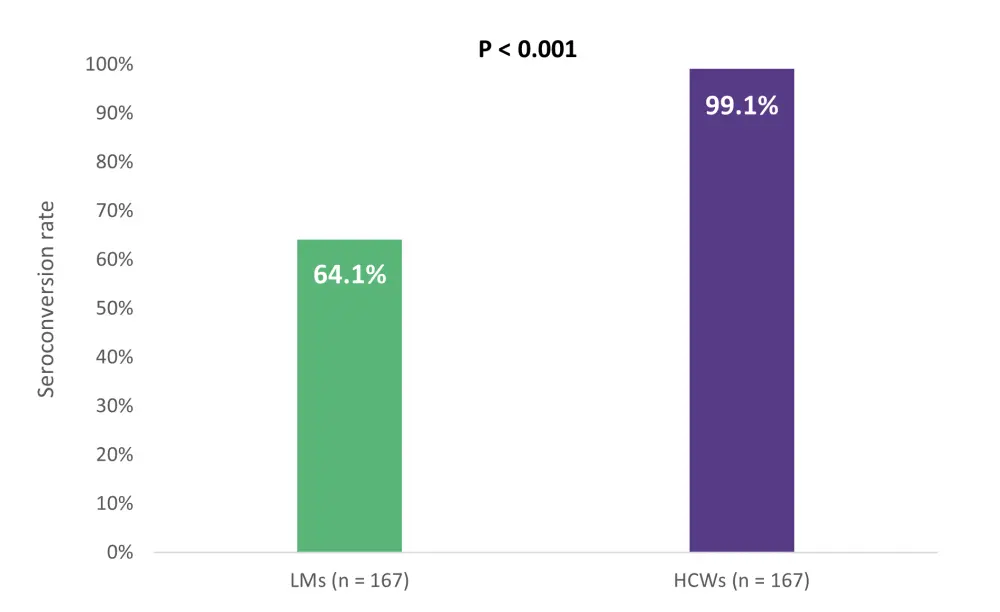
HCWs, healthcare workers; LMs, lymphoid malignancies.
*Adapted from Marasco, et al.1
On multivariate analysis, independent predictors of serological conversion included the type of treatment, type of diagnosis, absolute lymphocyte count, and immunoglobulin M levels (Table 2).
Table 2. Rate of seroconversion and antibody titer at two weeks after second dose*
|
95% CI, 95% confidence interval; OR, odds ratio. *Adapted from Marasco, et al.1 |
|||
|
Variable |
OR |
95% CI |
p value |
|---|---|---|---|
|
Diagnosis |
|||
|
Hodgkin lymphoma |
1 |
— |
<0.001 |
|
Aggressive B-cell lymphomas |
0.54 |
0.15–1.91 |
— |
|
Indolent B-cell lymphomas or B-cell |
1.33 |
0.36–4.93 |
— |
|
Multiple myeloma |
28.15 |
4.43–178.95 |
— |
|
T-cell lymphoma |
0.5 |
0.06–3.82 |
— |
|
Type of treatment |
|||
|
Other therapies |
1 |
|
<0.001 |
|
Anti-CD20 antibody plus chemotherapy |
0.07 |
0.02–0.22 |
|
|
Watch and wait or last therapy >12 |
2.84 |
0.96–8.36 |
— |
|
Absolute lymphocytic count |
|||
|
<800 cells/µL |
1 |
— |
0.001 |
|
≥800 cells/µL |
1.66 |
0.65–4.25 |
— |
|
Immunoglobulin M |
|||
|
<40mg/ dl |
1 |
— |
0.004 |
|
≥40 mg/dl |
4.31 |
1.52–12.24 |
— |
T-cell mediated immune response was assessed in 99 patients on active treatment and 99 healthy controls by measuring interferon-gamma (IFN-γ), interleukin-2 and tumor necrosis factor alpha levels (Table 3). A high proportion of patients with lymphoid malignancies achieved a T-cell mediated response, though it was lower than that of the healthcare worker control group. Patients with lymphoid malignancies also had a lower median level of IFN-γ and tumor necrosis factor alpha release compared with healthcare workers. In contrast, interleukin-2 levels were higher in patients with lymphoid malignancies compared with healthy controls.
Table 3. Comparison of T-cell mediated responses*
|
HCW, healthcare worker; IFN-γ; interferon gamma; IL-2, interleukin-2; LM, lymphoid malignancy; TNF-α, tumor necrosis factor-alpha. |
|||
|
|
Patients with LMs |
HCWs (n = 99) |
p value |
|---|---|---|---|
|
T-cell mediated |
86 |
100 |
<0.001 |
|
Median IFN-γ, U/ml |
179.5 |
309 |
<0.0001 |
|
Median TNF-α, pg/ml |
32.71 |
104.0 |
<0.0001 |
|
Median IL-2, pg/ml |
553.6 |
196.0 |
<0.0001 |
Notably, T-cell immune response was detectable in 47 (98%) seropositive patients and 38 (74%) seronegative patients. However, 13% of patients were defined as “double negative” with neither detection of a humoral or T-cell mediated response.
In summary, active lymphoid malignancies were confirmed to be a negative predictor of serological response to SARS-CoV-2 vaccination in line with previous research; however, a significantly increased serological response rate 2 weeks following the second dose emphasizes the importance of double dosing. Active treatment with anti-CD20 plus chemotherapy, CAR T-cell therapy, or novel oral agents in the 12 months prior to vaccination was associated with reduced serological response rates.
The use of B-cell depleting anti-CD20 agents produced an extremely low seroconversion rate, perhaps owing to the prolonged half-life of agents such as rituximab. Patients with multiple myeloma treated with immunomodulatory imide drugs had higher seroconversion (84%)—contradicting previous studies—which may be a result of a small cohort number and concomitant treatment.
Measurement of T helper cytokine release indicated alternative protection against infection, with cytokine release detected in 74% of seronegative patients; however, a notable fraction (13%) of patients had neither humoral or T-cell responses and may be at high risk for COVID-19 infection and related complications.
Liebers, et al.2
It has been demonstrated previously that patients treated with anti-CD20 plus chemotherapy had impaired humoral responses, and further data was published by Liebers, et al.2 who investigated seroconversion and T-cell mediated responses specifically for patients treated with these agents, either as a monotherapy or in combination with other agents.
The authors measured anti-SARS-CoV-2-S1 and anti-SARS-CoV-2-N antibody levels in patients without previous COVID-19 infection and assessed the T-cell response in a subset of patients. Patients with a known history of COVID-19, as well as those with detectable nucleocapsid protein antibodies (anti-SARS-CoV-2-N) were excluded.
Results
Patient characteristics are summarized in Table 4.
Table 4. Patient characteristics*
|
AlloHCT, allogeneic hematopoietic cell transplantation; AutoHCT, autologous hematopoietic cell transplantation; WBC, white blood cell count. |
|
|
Characteristic, % (unless otherwise stated) |
N = 80 |
|---|---|
|
Median age (range), years |
66 (23–88) |
|
Female |
38 |
|
Diagnosis |
|
|
Aggressive lymphomas |
40 |
|
Indolent lymphoma |
40 |
|
Chronic lymphocytic leukemia |
14 |
|
Nodular lymphocyte-predominant Hodgkin lymphoma |
5 |
|
Hairy cell leukemia |
1 |
|
Active treatment |
31 |
|
Treatment <12 months |
56 |
|
Last/current treatment |
|
|
Anti-CD20 + chemotherapy |
41 |
|
Anti-CD20 monotherapy |
29 |
|
Anti-CD20 + novel agents |
15 |
|
AutoHSCT following anti-C20 + |
8 |
|
Novel agents following anti-CD20 failure |
8 |
|
No prior alloHCT |
100 |
|
Median interval between first and second vaccination (range), |
42.0 (12.0–84.0) |
|
No second vaccination until data cut-off |
5 |
Following two vaccine doses, the overall seroconversion rate was 41%, a significant increase from the first dose (9%), confirming the importance of two doses in patients treated with immunosuppressive agents. Importantly, patients who had their last treatment >12 months prior to vaccination demonstrated higher seroconversion rates compared with those receiving last treatment within 3 to 12 months (p = 0.001) or <3 months (p < 0.001) (Figure 3).
Figure 3. Seroconversion rates dependent of the time interval of last anti-CD20 treatment to vaccination*
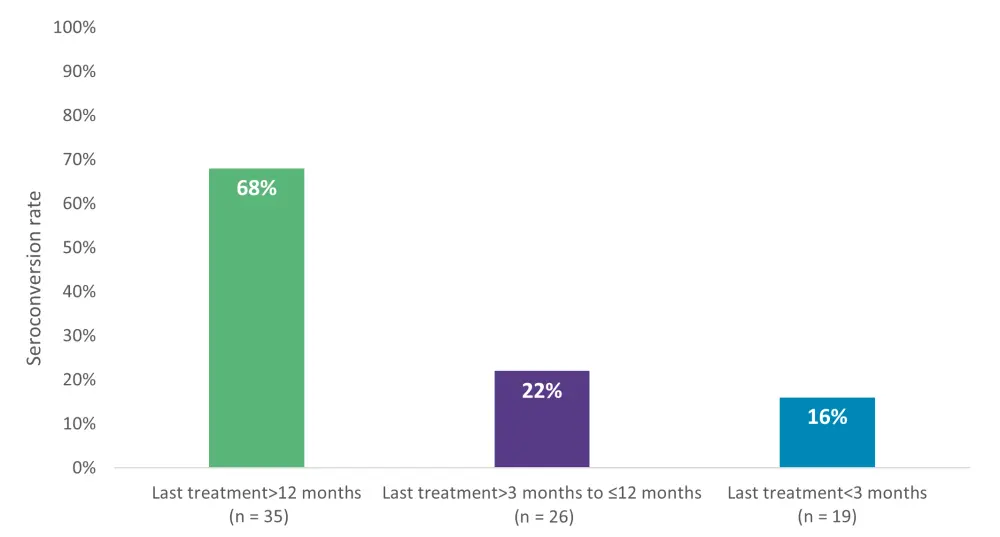
*Adapted from Liebers, et al.2
Notably, no statistically significant difference was reported when comparing seroconversion rates between patients receiving anti-C20 monotherapy or in combination with chemotherapy.
A multivariate analysis confirmed the interval from last anti-CD20 treatment to vaccination as an independent predictor of serological response (per 1 year; OR, 2.2; 95% CI, 1.3–4.7; p = 0.02) and revealed high CD4 cell counts (per 100 cells/µl; OR, 1.6; 95% CI, 1.2–2.3; p = 0.005) and age (per 10 years; OR, 0.5; 95% CI, 0.2–0.8; p = 0.008) as additional predictors.
T-cell responses were measured using IFN-γ levels in patients with available peripheral blood samples (n = 50), and these were compared with seven healthy vaccinated controls (Figure 4). The number of patients achieving seroconversion was comparable between patients and healthy controls. Half of the patients who did not seroconvert still had a positive T-cell response, providing further evidence of T-cell activity in patients with humoral suppression.
Figure 4. Percentage of patients achieving a T-cell response and T-cell response status in patients with or without seroconversion*
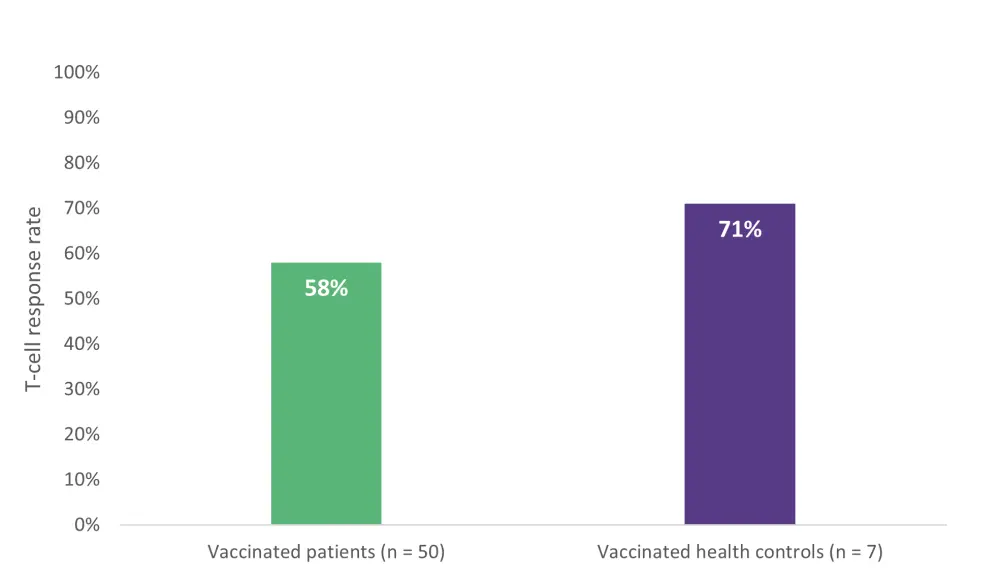
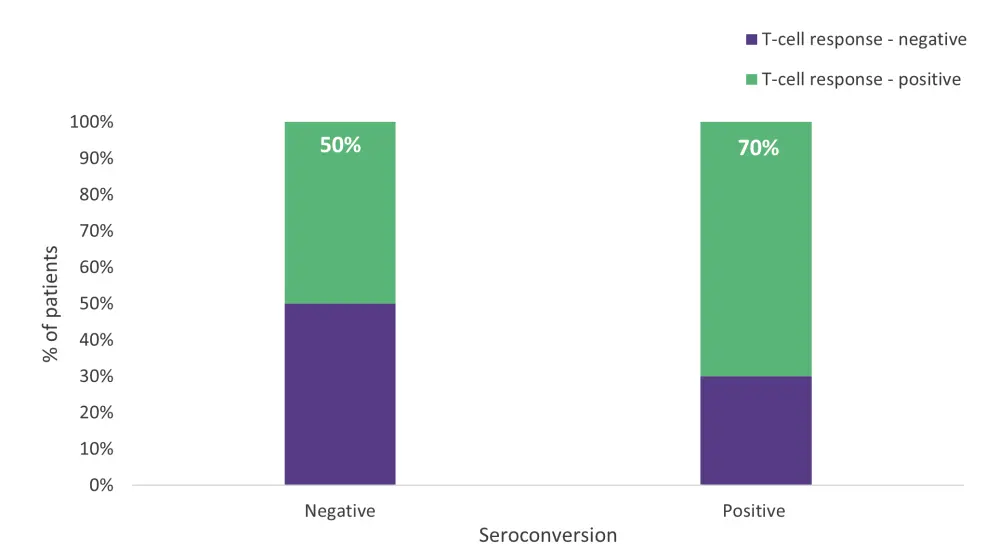
*Adapted from Liebers, et al.2
In contrast with serological response, T-cell mediated immune response was not affected by the time interval between last anti-CD20 treatment and first vaccine dose. When assessing T-cell responses following vaccination in patients with proven negative pre-vaccination T-cell responses (n = 20), the authors reported T-cell responses comparable to the entire cohort that were independent of seroconversion.
In summary:
- This study confirmed that anti-CD20 treatment is associated with reduced serological response, and patients with treatment within 12 months had significantly impaired responses.
- T-cell immunity was again demonstrated in a significant number of patients negative for serological response and was comparable with healthy controls.
- T-cell response appeared dependent of vaccination, understating their importance for this vulnerable population.
The limitations of this study included an overall small cohort sample set, and a small number of healthy controls used for comparison.
Conclusion
Data from both studies confirm the significant impact of lymphoid malignancies on serological response. In addition, they demonstrated a negative association of anti-CD20 treatment, either in combination with chemotherapy or as a monotherapy. Furthermore, an independent negative predictor was the time interval of last anti-CD20 treatment to vaccination. Second doses appeared to improve the rate of seroconversion; however, further data is needed to evaluate whether a booster dose will further improve the seroconversion rate.
A key takeaway from these studies was the presence of T-cell immune responses in a significant number of patients with lymphoid malignancies who failed to seroconvert. Data from Liebers, et al.2 appeared to show that the T-cell response was dependent on vaccination, providing evidence for the importance of vaccination in this population, with T-cell mediated responses perhaps providing compensatory protection against infection in those lacking humoral activity.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average vein-to-vein time when treating patients with DLBCL with a reimbursed CAR T-cell therapy (from apheresis to infusion)?

