All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The lym Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the lym Hub cannot guarantee the accuracy of translated content. The lym and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Patient preferences for the treatment of Waldenstrom's macroglobulinemia
The management of Waldenstrom's macroglobulinemia (WM) has advanced in recent years, as previously reported on the Lymphoma Hub, with current treatments varying by efficacy, toxicity profile, treatment duration, type of agent, and route of administration. Currently, there is no consensus on a preferred treatment approach; therefore, a better understanding of patient preferences would aid physicians and patients in shared decision-making. Information on patient preferences may also help direct future clinical trials in WM and influence healthcare decision-making by engaging patients across different stages of clinical drug development and care management.
Discrete choice experiments (DCEs) are a quantitative way of eliciting preferences by asking respondents to make choice from two or more alternatives. Below, we summarize the findings from a DCE study by Amaador, et al.,1 published in Cancer Medicine evaluating patient preferences in the management of WM.
Method
This was a DCE using a mixed-method approach to identify and select attributes and levels to understand the preferences of patients with WM and their caregivers. This included:
- a graphically visualized subset of 16 hypothetical treatments;
- a pilot DCE that informed the definitive experimental DCE design included in the survey;
- an online or paper-based survey developed in Dutch, with the first part focused on socioeconomic and clinical characteristics and the second part on the detailed explanation of the DCE task; and
- a modified survey completed by patients’ caregivers.
The five attributes included in the questionnaire are listed in Figure 1.
Figure 1. Attributes and levels used in the DCE*
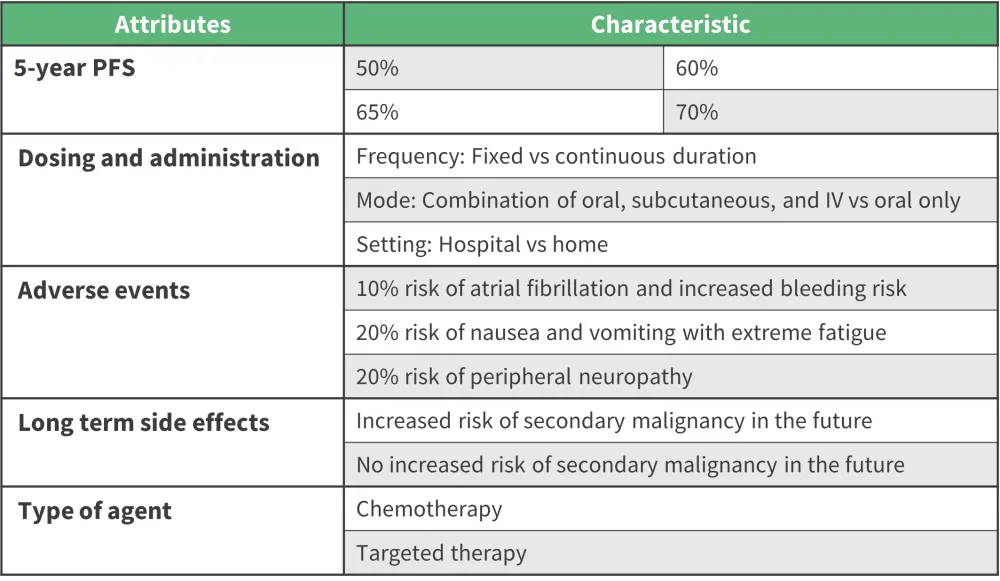
IV, intravenous; PFS, progression-free survival.
*Adapted from Amaador, et al.1
Results
Baseline characteristics
A total of 214 patients with WM completed the questionnaire and were included in the analysis. The median age of respondents was 67 years of age and 54% were male. Regarding treatment, 56% of respondents were previously treated, 25% were treatment naïve and 19% of respondents were being treated at the time of the survey (Table 1).
Table 1. Baseline characteristics*
|
DCE, discrete choice experiment. |
|
|
Characteristics (n/N), % (unless otherwise |
Patients completing DCE (N = 214) |
|---|---|
|
Time since diagnosis (range), years |
6.9 (0.1–48) |
|
High educational level† |
55 (115/209) |
|
Disease status |
|
|
Wait and see |
31 (66/211) |
|
Remission |
39 (83/211) |
|
Progression |
10 (20/211) |
|
Currently treated |
20 (42/211) |
|
Previously treated at time of questionnaire completion |
|
|
Yes |
58 (122/212) |
|
No |
28 (60/212) |
Patient preferences
The average marginal effects of all attributes except one (atrial fibrillation/increased risk of bleeding) were statistically significant (p < 0.001). The 5-year progression free survival (PFS) was the most important attribute for making treatment decisions followed by the risk of secondary malignancies in the future (Table 2).
Table 2. Attributes and probability of choosing a treatment*
|
CI, confidence interval; IV, intravenous; PFS, progression free survival; SC, subcutaneous. |
||
|
Attribute (95% CI), % |
Increase in probability of choosing a treatment |
Decrease in probability of choosing a treatment |
|---|---|---|
|
Increase in 5-year PFS from 50% to 70% |
26 (23–30) |
— |
|
Decrease in risk of future malignancies from high risk to low risk |
22 (18–27) |
— |
|
Increased risk of neuropathy |
— |
11 (7 to 14) |
|
Treatment contained targeted therapy |
8 (6–10) |
— |
|
Fixed-duration treatment with an IV/SC administration at the hospital versus continuous daily oral treatment at home |
8 (5–10) |
— |
Patients were willing to trade treatment efficacy to receive a treatment with certain attributes, such as no increased risk of secondary malignancy, a fixed duration treatment with intravenous or subcutaneous administration at the clinic, or a treatment containing targeted therapy. On the other hand, patients were only willing to accept a treatment with atrial fibrillation or neuropathy as a side effect if they received a 1.4% and 7.2% increase in treatment efficacy, respectively (Figure 2).
Figure 2. Willingness to trade efficacy for other attributes*
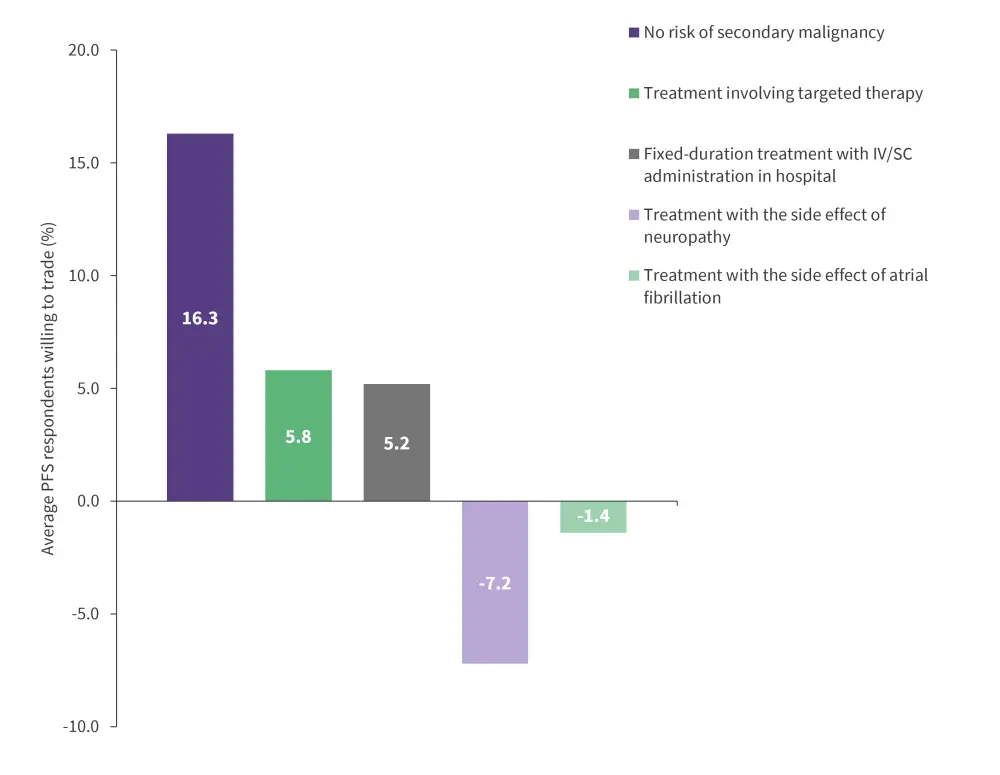
IV, intravenous; PFS, progression free survival; SC, subcutaneous.
*Adapted from Amaador et al.1
Patients rated the difficulty level of the survey with a median of 5.4 on a 10-point scale. Based on the interviews in the pilot study, patients found the survey questions emotionally challenging rather than difficult to understand.
Caregiver preferences
For caregivers, only 3 out of 5 attributes had a significant influence on their treatment preferences. These included 5-year PFS, risk of secondary malignancy, and type of agent; this was consistent with patients’ preferences (p = 0.22).
Conclusion
This Dutch DCE study, the first of its kind, showed that long PFS and low risk of secondary malignancies were the most important attributes for patients choosing treatment for WM. Neuropathy was the adverse event patients most wanted to avoid, highlighting the need to identify active regimens that are not neurotoxic, and patients showed a preference for fixed-duration intravenous or subcutaneous treatment in a hospital setting over continuous daily oral treatment at home.
The authors consider these findings valuable for directing future clinical trials, developing novel drugs, informing clinical guidelines, and influencing healthcare decision-making. Strengths of this study include selection of attributes and levels by WM experts and patients, large sample size, and a good response rate to the survey. However, the findings may be limited by selection bias, uncertainty regarding the difficulty of completing the survey, non-generalizability of the findings worldwide due to differences in culture, infrastructure, and healthcare systems.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average time to secure a reimbursed CAR T-cell therapy manufacturing slot for patients with DLBCL (from decision to treatment with a CAR T-cell therapy)?

