All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The lym Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the lym Hub cannot guarantee the accuracy of translated content. The lym and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
AACR 2017 | Poster 3631/4 – Delivery of a CD20 transferrin receptor VNAR bispecific antibody to the brain for Central Nervous System Lymphoma
At the American Association for Cancer Research (AACR) annual meeting in Washington, DC, USA, on Tuesday 4th April, a poster session titled “BITES Bispecifics and Checkpoints” took place.
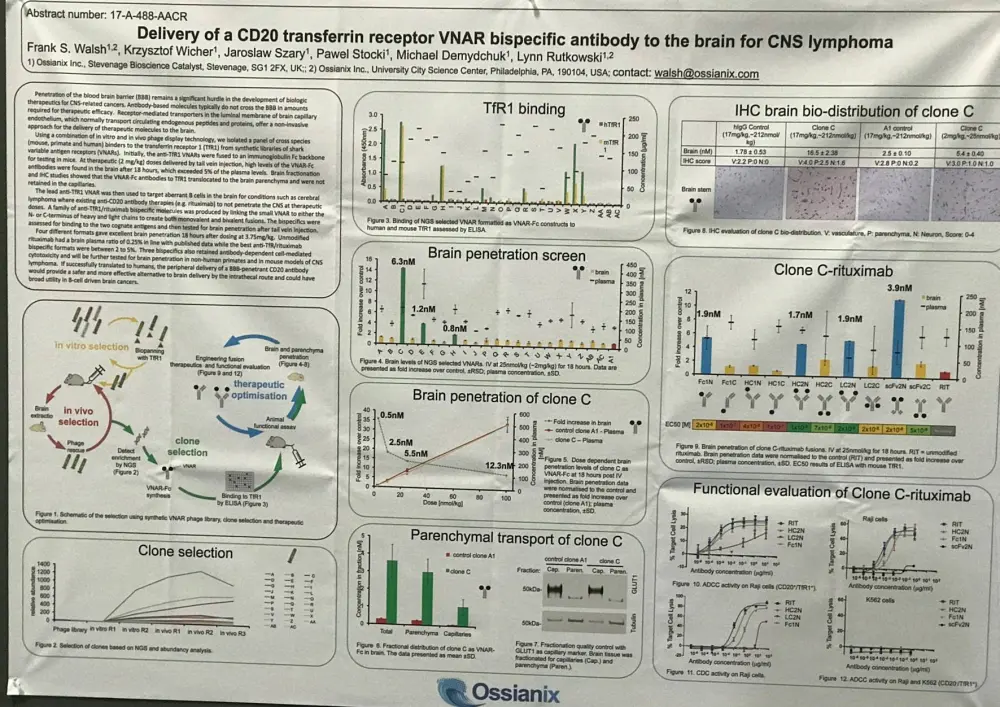
One of the posters on display (3631 / 4) was titled “Delivery of a CD20 transferrin receptor VNAR bispecific antibody to the brain for CNS lymphoma” by Frank S. Walsh from Ossianix, Philadelphia, PA, and colleagues.
Currently, the challenge facing the development of biologic therapies for CNS-related cancers is getting the agent to cross the blood brain barrier.
This group used in vitro and in vivo phage display technology; they identified a panel of cross species (rodent, primate, and human) binders to the Transferrin Receptor 1 (TfR1) from synthetic libraries of shark Variable Antigen Receptors (VNARs).
Key Highlights:
- To begin with, the anti-TfR1 VNARs were attached to an Ig Fc backbone for testing in mice
- When therapeutic doses (2mg/kg) were administered via tail vein injection, high levels of VNAR-Fc antibodies were reported in the brain after 18 hours, >5% of the plasma levels
- Brain fractionation studies indicated that VNAR-Fc antibodies to TfR1 translocated to the brain parenchyma and were not retained in capillaries
- The lead anti-TfR1 VNAR was used to target aberrant B-cells in the brain for diseases like cerebral Lymphoma
- A family of anti-CD20/TfR1 bispecific molecules were produced; small VNAR were linked to either the N- and C- terminus of heavy and light chains to generate monovalent and bivalent fusions
- The anti-CD20 agent used was rituximab
- 18 hours after dosing at 3.75mg/kg, ≥4 different formats gave outstanding brain penetration
- Unmodified rituximab brain plasma ratio = 0.25; the best anti-TfR1-rituximab bispecific formats = between 2 to 5%
- The bispecific formats maintain Antibody-Dependent Cell Cytotoxicity (ADCC)
The poster concluded by stating that these bispecific formats will be investigated further for brain penetration in non-human primates and in CNS Lymphoma rodent models.
If these results are successfully replicated in larger animals, the delivery of the bispecific CD20/TfR1 antibody would present as a safer and more effective substitute to intrathecal delivery of CD20 antibody and could have wide range of uses to treat and manage B-cell driven brain cancers.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average time to secure a reimbursed CAR T-cell therapy manufacturing slot for patients with DLBCL (from decision to treatment with a CAR T-cell therapy)?

