All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Phase II study of lenalidomide and R-CHOP in patients with diffuse large B-cell lymphoma
Diffuse large B-cell lymphoma (DLBCL) is recognized to be a molecularly heterogenous malignancy. Of the three main subtypes, the activated B-cell-like (ABC) subtype has the poorest prognosis historically. However, the ABC subtype has responded better than the germinal center B-cell-like (GCB) subtype to treatment with lenalidomide in previous studies.
Therefore, the phase II ECOG-ACRIN E1412 study (NCT01856192) was set up in the US to test how patients with DLBCL responded to treatment with R-CHOP compared with R-CHOP plus lenalidomide (R2CHOP). The results were recently published in the Journal of Clinical Oncology by Grzegorz Nowakowski and colleagues, and the impact on different DLBCL subtypes was analyzed.1
Study design
A total of 280 patients were eligible for this study and were randomly assigned to each treatment arm. The treatment regimens for the two arms are shown in Figure 1.
Figure 1. Study treatment schedule: 1 cycle of a 6-cycle schedule1
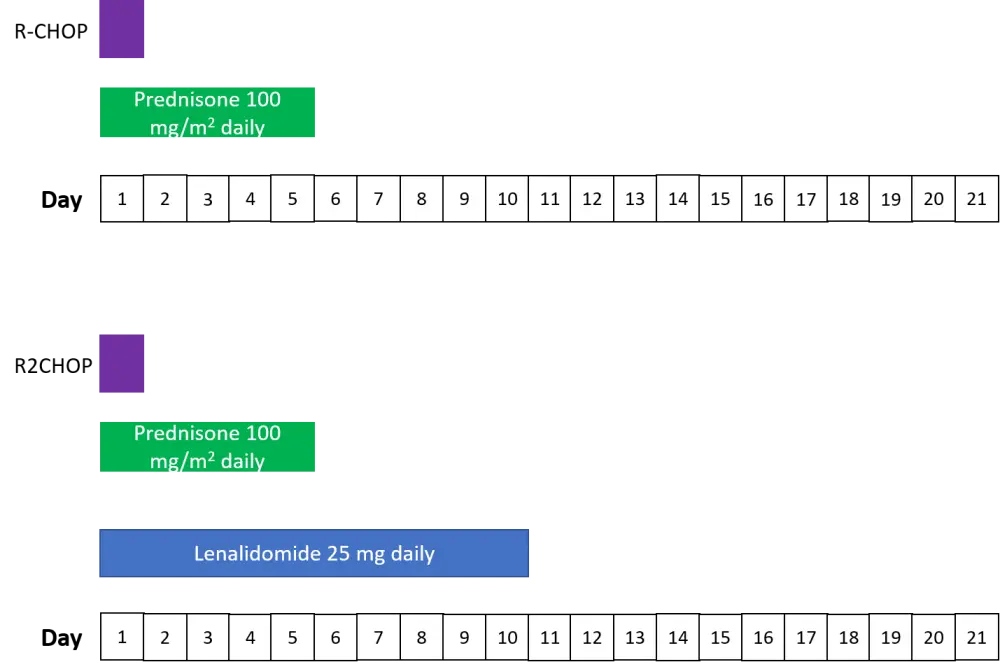
R-CHOP: rituximab 375 mg/m2, cyclophosphamide 750 mg/m2, doxorubicin 50 mg/m2, vincristine 1.4 mg/m2, and prednisone 100 mg/m2; R2CHOP, R-CHOP + lenalidomide.
Eligibility criteria:
- ≥ 18 years with newly diagnosed, untreated DLBCL that had been histologically assessed
- Stage II bulky to IV disease
- Eastern Cooperative Oncology Group performance status (ECOG PS) of 0−2
- International Prognostic Index of 2−5
Exclusion criteria:
- Known central nervous system lymphoma
- Deep vein thrombosis or emboli or thrombophilia history (unless willing to be on full anticoagulation treatment) if assigned to the R2CHOP arm
- Acquired immunodeficiency syndrome-related conditions
Key points
Baseline characteristics were similar between the two treatment groups. Most patients in this study had high-risk disease, with > 96% Stage III or IV, and over 60% with an ECOG PS of 1−2. The ABC subtype made up 40% of the 234 patients that were evaluated for cell of origin, and their characteristics are listed separately from the main cohort (Table 1).
Table 1. Patient baseline characteristics in the two treatment arms and for the ABC subtype1
|
ABC, activated B-cell-like; DLBCL, diffuse large B-cell lymphoma; dx, diagnosis; ECOG, Eastern Cooperative Oncology Group; GCB, germinal center B-cell-like; IPI, International Prognostic Index; PS, performance status; R-CHOP, rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone; rx, treatment. |
||||
|
Characteristic |
All (N = 280) |
ABC-DLBCL (n = 94) |
||
|---|---|---|---|---|
|
R2CHOP (n =145) |
R-CHOP (n = 135) |
R2CHOP (n = 44) |
R-CHOP (n = 50) |
|
|
Age, years median (range) |
67 (24−88) |
66 (37−92) |
67 (24−88) |
67 (37−80) |
|
Time dx to rx, days median (range) |
21 (1−111) |
19 (2−134) |
21 (5−83) |
19 (2−134) |
|
Sex, male (%) |
65 |
56 |
68 |
42 |
|
Stage III and/or IV (%) |
97 |
96 |
100 |
97 |
|
Bulky disease ≥ 7 cm (%) |
41 |
43 |
30 |
44 |
|
Extra nodal sites ≥ 2 (%) |
45 |
47 |
40 |
32 |
|
ECOG PS (%) |
||||
|
0 |
39 |
35 |
41 |
34 |
|
1−2 |
61 |
65 |
59 |
66 |
|
IPI (%) |
||||
|
2 |
33 |
34 |
36 |
44 |
|
3 |
43 |
42 |
43 |
40 |
|
4−5 |
23 |
24 |
20 |
16 |
|
Cell of origin*, n |
||||
|
ABC |
44 |
50 |
|
|
|
GCB |
66 |
56 |
|
|
|
Unclassified |
9 |
9 |
|
|
|
Unknown |
26 |
20 |
|
|
Efficacy
With a median follow-up of 3 years, the overall response rates (ORR), and complete response rates (CR) were as follows:
ORR
- R2CHOP: 97%
- R-CHOP: 92% (p = 0.06)
CR
- R2CHOP: 73%
- R-CHOP: 68% (p = 0.43)
The primary endpoint of progression-free survival (PFS) was met in this study; PFS rates were as follows for R2CHOP versus R-CHOP:
- 1-year PFS: 84% vs 73%
- 2-year PFS: 76% vs 69%
- 3-year PFS: 73% vs 62%
Overall survival (OS) was increased in the R2CHOP group compared with the R-CHOP group, with a 3-year OS of 83% vs 75% (one-sided p = 0.05), respectively.
The R2CHOP group showed a 34% decrease in risk of death or progression vs the R-CHOP group (HR 0.66, one-sided 90% CI < 0.88; one-sided p = 0.03).
Analysis by cell of origin showed that R2CHOP improved outcomes in patients with an ABC subtype (n = 94; HR 0.64; one-sided 90% CI upper limit, 1.01; two-sided 95% CI, 0.31−1.29; one-sided p = 0.1). The HR in the GCB treatment group (n = 122) was not as reduced as in the ABC cohort at 0.82 (one-sided 90% CI upper limit, 1.27; two-sided 95% CI, 0.43−1.59).
Safety
In the R2CHOP arm there was one case of Grade V lung infection recorded. In the R-CHOP cohort four Grade V cases were recorded: one Grade V case of fatigue, two Grade V sepsis cases, and one Grade V lung infection.
The adverse events (AEs) experienced by patients were similar between groups, with anemia, neutropenia, and thrombocytopenia being the most frequently recorded Grade 3–4 AEs (Table 2).
Significant differences between R2CHOP and R-CHOP arms were found for the following Grade ≥ 3 AEs:
- anemia (29% vs 20%, p = 0.003)
- febrile neutropenia (25% vs 14%, p = 0.003)
- thrombocytopenia (34% vs 13%, p = 0.001)
- diarrhea (6% vs 1%, p = 0.005)
- electrolyte abnormalities (5% vs 2%, p = 0.06)
Table 2. Grade 3–4 adverse events for R2CHOP and R-CHOP treatment arms1
|
R-CHOP, rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone. R2CHOP, lenalidomide + R-CHOP. |
||||
|
Adverse event (%) |
R2CHOP (n = 166) |
R-CHOP (n = 171) |
||
|---|---|---|---|---|
|
Grade 3 |
Grade 4 |
Grade 3 |
Grade 4 |
|
|
Anemia |
25 |
4 |
18 |
2 |
|
Febrile neutropenia |
20 |
5 |
11 |
1 |
|
Fatigue |
10 |
— |
6 |
— |
|
Sepsis |
— |
7 |
— |
4 |
|
Lung infection |
5 |
1 |
4 |
— |
|
Neutropenia |
9 |
51 |
12 |
42 |
|
Thrombocytopenia |
14 |
20 |
4 |
9 |
|
Hypokalemia |
5 |
1 |
1 |
1 |
|
Hyponatremia |
5 |
— |
4 |
— |
|
Hypophosphatemia |
5 |
— |
1 |
— |
|
Generalized muscle weakness |
5 |
— |
2 |
— |
|
Rash maculopapular |
2 |
— |
— |
— |
|
Thromboembolic event |
3 |
1 |
2 |
— |
The percentage of patients that completed six cycles of therapy was very similar between treatment arms (86% and 85% in the R2CHOP and R-CHOP arms, respectively). Treatment discontinuation occurred in 8% of patients receiving R2CHOP and 3% of patients receiving R-CHOP alone.
During the study there were nine treatment-related deaths, two in the R2CHOP arm (both due to lung infection) and seven in the R-CHOP arm (five due to sepsis or febrile neutropenia, one due to lung infection, and one due to adult respiratory distress syndrome).
Conclusions
The ECOG-ACRIN E1412 study demonstrated improved outcomes for patients with newly diagnosed DLBCL upon adding lenalidomide to R-CHOP, irrespective of the cell of origin subtype, although the superior OS and PFS observed for the R2CHOP group compared with the R-CHOP group was most notable for patients with ABC DLBCL. The safety profiles were similar between treatment groups and in line with what is expected for R-CHOP protocols. These results support further studies of lenalidomide in the frontline treatment of DLBCL.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

