All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Tandem CD20–CD19 CAR T-cell therapy for relapsed B-cell malignancies
The novel chimeric antigen receptor (CAR) T-cell therapies targeting CD19 achieved unprecedented survival outcomes in heavily pretreated patients with advanced B-cell malignancies. However, some patients still relapse, mostly with CD19-negative disease, reducing considerably their treatment options left at this stage.
Tandem CAR T cells aim to target two essential antigens widely presented in B-cell non-Hodgkin lymphoma (NHL) and chronic lymphocytic leukemia (CLL): CD19 and CD20. Dual targeting might improve the clinical outcomes and overcome antigen-loss relapses seen so far with singly targeted CD19 CAR T-cell therapies.
Nirav N. Shah and colleagues published recently in Nature Medicine the first experience with an onsite manufactured tandem CD19–CD20 CAR T cell for patients with relapsed B-cell malignancies.1 Below there is a summary of the study results.
Study design
- This is a first-in-human phase I trial to evaluate the safety and feasibility of tandem CD20–CD19 4-1BB–CD3ζ lentiviral (LV20.19) CAR T-cell therapy for patients with relapsed/refractory B-cell malignancies (NCT03019055).
- A total of 26 patients were enrolled, of which 22 achieved the target dose. The key selection criteria were:
- Measurable B-cell NHL or CLL, treated with at least two prior lines of therapy.
- CD19 or CD20 positive disease (≥ 5% by immunohistochemistry or flow cytometry) in the most recent biopsy.
- CD3-positive T-cell count of at least 50/mm3.
- Karnofsky performance score ≥ 70.
- No active central nervous system involvement.
- Prior CAR T-cell therapy was permitted if patients remained positive for CD19 or CD20.
- After apheresis and lymphodepletion (30 mg/m2 fludarabine for 3 days and 500 mg/m2 cyclophosphamide for 1 day), patients were divided into three cohorts:
- Cohort 1: dose escalation at dose levels 2.5 × 105 cells/kg, 7.5 × 105 cells/kg, and a target dose of 2.5 × 106 cells/kg, administered over 2 days (30–70%).
- Cohort 2: expansion cohort with dose selected from cohort 1, split into a 2-day infusion.
- Cohort 3: expansion cohort with selected dose, single infusion.
LV20.19 CAR T-cell manufacturing
- The CliniMACS Prodigy is a new closed-system device that enables the production of CAR T cells locally.2 Tandem LV20.19 CAR T cells were manufactured onsite at the Medical College of Wisconsin.
- Once adjusted and tested appropriately, it can reduce manufacturing timelines (14 days), and patients can receive fresh CAR T cells (non-cryopreserved).
- In the cell cultures, a mean of 17.4% were CAR T cells (range, 7.4–28.0%), with a mean CD4:CD8 ratio of 3:1 at the time of cell collection.
- The final product reached an average of 5.76 × 108 LV20.19 CAR T cells (range, 2.22–12.8 × 108).
Results
- Patients' baseline characteristics are summarized in Table 1.
Table 1. Patient characteristics at the time of CAR T-cell infusion1
|
AA, African American; allo, allogeneic; auto, autologous; CAR, chimeric antigen receptor; CLL, chronic lymphocytic leukemia; DLBCL, diffuse-large B-cell lymphoma; EA, East Asian; FL, follicular lymphoma; MCL, mantle cell lymphoma; SCT, stem cell transplant. |
|
|
Characteristic |
Patients (N = 22) |
|---|---|
|
Median age, years (range) |
57 (38–72) |
|
Race, % |
|
|
European ancestry |
86 |
|
AA, EA, or Native American |
14 |
|
Lymphoid malignancy, % |
|
|
DLBCL |
50 |
|
MCL |
32 |
|
CLL |
14 |
|
FL |
4 |
|
Median lines of prior therapy (range) |
4 (2–12) |
|
Refractory to the last line of therapy, % |
82 |
|
Prior auto-SCT, % |
37 |
|
Prior allo-SCT, % |
14 |
|
Patients infused at each dose level, % |
|
|
2.5 × 105 cells/kg |
14 |
|
7.5 × 105 cells/kg |
14 |
|
2.5 × 106 cells/kg |
73 |
|
Split infusion, % |
73 |
|
Non-cryopreserved cell product infused, % |
68 |
- Tandem LV20.19 CAR T cells achieved an overall response rate (ORR) of 82% after 28 days from infusion, including high complete response (CR) rates across all subtypes (Figure 1).
- Higher rates and better responses were achieved in the target dose level cohort and in patients infused with fresh cells; although the difference is not statistically significant (p = 0.08), it is important to note that these patients also presented a higher expansion of the CAR T-cell product.
- After a median follow-up of 10.1 months, the median duration of response in patients achieving a CR was not reached.
- To date, nine (40.9%) patients progressed after initial response to LV20.19 CAR T cells; four of them were late relapses (≥ 6 months after infusion). Three patients progressed again after retreatment with the cryopreserved product.
Figure 1. Response rates at Day 28 after infusion of LV20.19 CAR T cells by type of B-cell NHL1
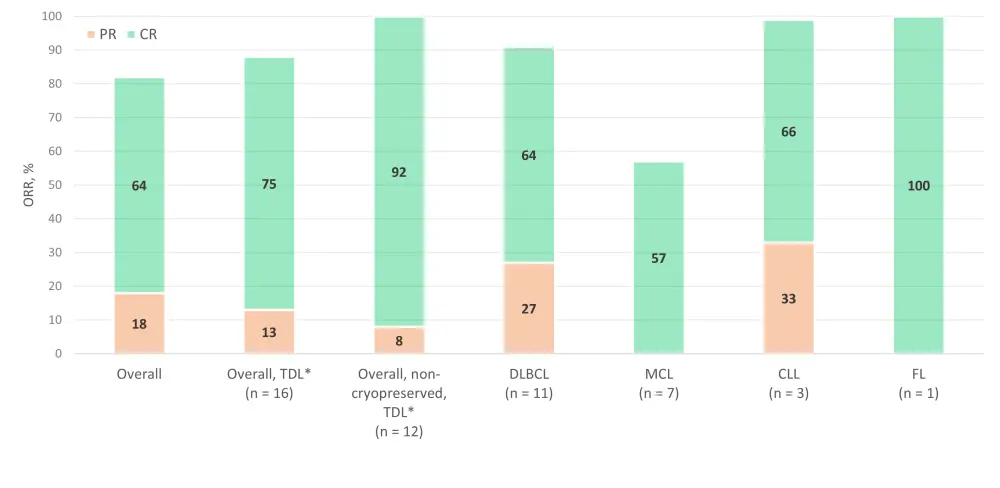
CAR, chimeric antigen receptor; CLL, chronic lymphocytic leukemia; CR, complete response; DLBCL, diffuse-large B-cell lymphoma; FL, follicular lymphoma; MCL, mantle cell lymphoma; NHL, non-Hodgkin lymphoma; ORR, overall response rate; PR, partial response; TDL, target dose level.
*2.5 × 106 cells/kg.
- Patients who were previously exposed to an anti-CD19 CAR T-cell therapy (n = 5) presented lower manufacturing success. Three were infused with the tandem CAR T cells but progressed by Day 28.
- Split and single infusion cohorts did not differ in response rates or adverse event incidence.
- Median overall survival was 20.3 months.
Predicting response to LV20.19 CAR T cells
- Baseline CD19 and CD20 expression was not associated with response to LV20.19 CAR T cells.
- Responders presented a peak of CAR T-cell expansion in peripheral blood at 7–12 days and were undetectable by Day 90 post-infusion.
- Higher levels of naïve and central memory T cells in the apheresis product were detected in patients later achieving a partial response or CR at the higher dose level.
- Five patients with progressive disease were infused with a lower dose level than 2.5 × 106 cells/kg.
- Biopsies after progression to LV20.19 CAR T cells show persistence of CD19 expression.
Safety
- There were no dose-limiting toxicities reported in the dose-escalation cohorts, although one patient in the expansion cohort experienced a dose-limiting toxicity at 2.5 × 106 cells/kg, requiring urgent hospitalization due to Grade 4 cytokine release syndrome (CRS) and neurotoxicity.
- The most frequent Grade ≥ 3 adverse events (AEs) with the higher CAR T-cell dose were cytopenia, hypoxia, and neurotoxicity. See Table 2 for a detailed list.
- Considering all dose levels, a total of 14 patients (64%) experienced CRS, and seven patients (32%) reported a neurotoxic event of any grade at a median time of 5 and 6 days from infusion, respectively.
- There were no deaths associated with LV20.19 CAR T-cell therapy.
Table 2. Most frequently reported AEs with LV20.19 CAR T cells at dose 2.5 × 106 cells/kg (n = 16)1
|
AE, adverse event; ALT, alanine aminotransferase; CAR, chimeric antigen receptor; CRS, cytokine release syndrome; WBC, white blood cell. |
||
|
AE reported in > 25% of patients |
All Grades, % |
Grade ≥ 3, % |
|---|---|---|
|
Lymphocytopenia |
88 |
88 |
|
Decrease in WBCs |
88 |
75 |
|
Neutropenia |
81 |
69 |
|
Anemia |
75 |
44 |
|
CRS |
75 |
6 |
|
Hypocalcemia |
63 |
6 |
|
Thrombocytopenia |
56 |
31 |
|
Increased creatinine |
44 |
6 |
|
ALT increased |
38 |
6 |
|
Neurotoxicity |
31 |
19 |
Conclusion
This first-in-human study reported the feasibility and safety of dual-targeting CD19 and CD20 with the tandem CAR T-cell therapy LV20.19 for patients with relapsed or refractory B-cell malignancies. With the possibility of onsite manufacturing, timelines were optimized, and it was possible to infuse non-cryopreserved product at 2.5 × 106 cells/kg, which led to a potentially better expansion of the CAR T cells, hence, a possibility to improve the clinical outcomes. This local procedure had an 85% success rate, including the 100% of patients naïve to CAR T cells, but needs to be further investigated.
In light of the promising response rates, the phase I/II trial with LV20.19 in B-cell NHL and mantle cell lymphoma has already started to recruit participants (NCT04186520), including patients previously treated with CD20-directed therapy and anti-CD19 CAR T-cell therapy.
A similar strategy with the same device has been tested in the University Hospital Cologne, Germany, by Peter Borchmann and colleagues. The initial results of the MB-CART2019.1 tandem CAR T-cell therapy in relapsed/refractory B-cell NHL were presented recently at the 62nd American Society of Hematology (ASH) Annual Meeting and Exposition. Watch below a summary of the phase I trial results, which, so far, are consistent with the ones reported with LV20.19.
Results from the phase I study of a novel CD19/CD20 tandem CAR T-cell therapy in R/R B-cell NHL
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What is the primary reason you use bridging therapy in patients with DLBCL awaiting CAR T-cell therapy?

