All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit the Lymphoma Coalition.
The Lymphoma Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the Lymphoma Hub cannot guarantee the accuracy of translated content. The Lymphoma Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The Lymphoma & CLL Hub is an independent medical education platform, sponsored by AbbVie, BeOne Medicines, Miltenyi Biomedicine, Nurix Therapeutics, Roche, Sobi, and Thermo Fisher Scientific and supported through educational grants from Bristol Myers Squibb, Eli Lilly, and Pfizer. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View lymphoma & CLL content recommended for you
Three-year follow-up of KTE-X19 in R/R mantle cell lymphoma: ZUMA-2 trial
Brexucabtagene autoleucel (KTE-X19) is an autologous anti-CD19 chimeric antigen receptor (CAR) T-cell product, currently approved by the U.S. Food and Drug Administration (FDA) for the treatment of relapsed/refractory (R/R) mantle cell lymphoma (MCL). ZUMA-2 (NCT02601313) is a phase II study of KTE-X19 in patients with heavily pretreated MCL. The Lymphoma Hub has previously reported on the study design and phase II results of the ZUMA-2 trial. Below, we provide a summary of the 3-year follow-up results as published by Wang, et al., in Journal of Clinical Oncology in 2022.1
Study design
As previously reported, 74 patients were enrolled in the trial and 68 patients received treatment with KTE-X19. At the data cutoff, the median follow-up time was 35.6 months. Several high-risk sub-groups were present in the patient population, including patients with blastoid variant, high Ki-67 proliferation index, tumor protein p53 gene mutation16 or high p53 and disease progression within 24 months postdiagnosis. The primary endpoint was the objective response rate (ORR) assessed by independent radiologic review committee. Secondary endpoints were duration of response (DOR), progression-free survival (PFS), overall survival (OS), adverse event (AE) incidence, blood CAR T-cell levels, and serum cytokine levels. Minimal residual disease (MRD) was assessed as an exploratory endpoint. Post-hoc assessment of subgroups was also performed based on prognostic features.1
Results
Efficacy1
- In the all-treated population, ORR was 91% (95% confidence interval [CI], 81.8–96.7). Further details on the complete response (CR) and partial response (PR) are presented in Figure 1.
- Overall, 25 patients achieved CR or PR after initial stable disease
- Median DOR was 28.2 months (46.7 months for CR and 2.2 months for PR)
- At data cut-off, 37% of patients remained in ongoing response (all with CR)
- Median PFS and OS were 25.8 months and 46.6 months, respectively
- For those who achieved a CR, PR, and no response the median PFS was 48, 3.1, and 2.3 months, respectively
Figure 1. Responses in the all-treated population*
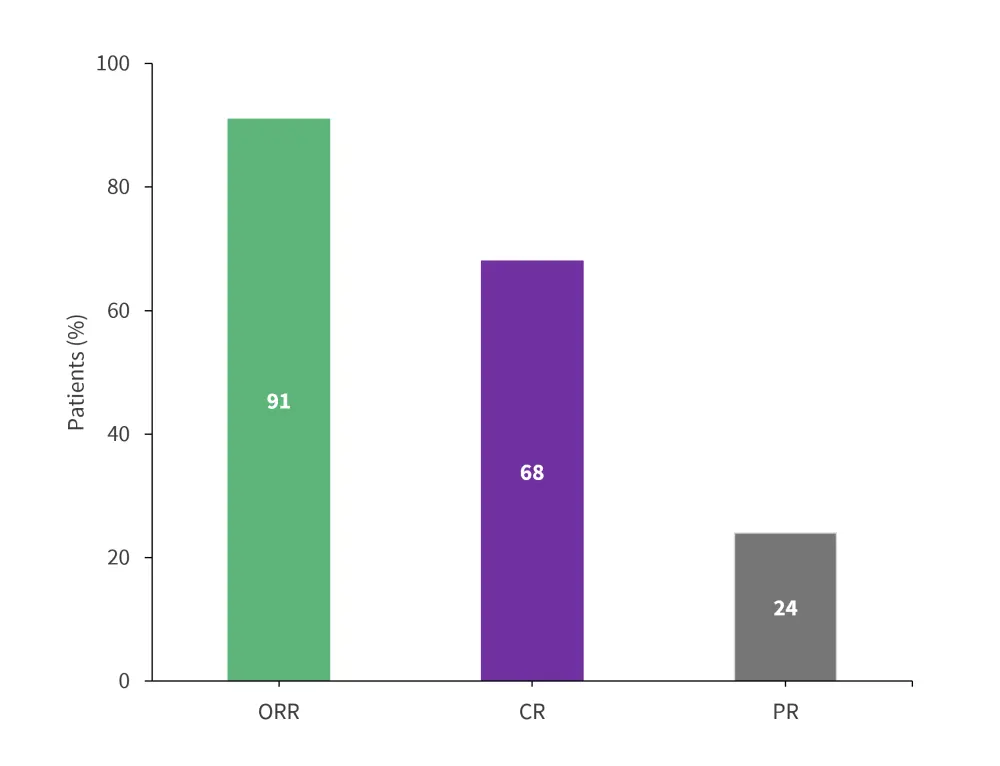
CR, complete response; ORR, objective response rate; PR, partial response.
*Adapted from Wang, et al.1
At 6 months, 79% of MRD-assessable patients were MRD-negative (ORR, 100%). At the data cutoff, median DOR, PFS, and OS were 6.1, 7.1, and 27 months among the MRD-positive patients, respectively, and MRD negativity at months 3, 6, and 9 was associated with durable response.
In the intention-to-treat population, ORR was 84% (CR, 62%; PR, 22%) and median PFS and OS were 24 months and 47.4 months, respectively. Response rates were stable among prespecified subgroups of prior Bruton’s tyrosine kinase inhibitor exposure or high-risk characteristics. Blood CAR T-cell levels were higher at a median of 15 days post-infusion, with median concentrations plateauing at 0.19–0.34 cells/µL from Month 6–18.
Safety1
No new safety signals were reported in the long-term follow-up of this trial, with only 3% of all adverse events of interest occurring since the previous data cutoff. AEs of any grade and ≥Grade 3 occurred in 26% and 21% of patients, respectively, and the most frequent AEs included cytopenia (19%), infections (10%), and neurologic events (3%).
Conclusion
This longer-term follow-up of ZUMA-2 study demonstrated the durable long-term effectiveness and safety of KTE-X19 in patients with R/R MCL. In addition, KTE-X19 showed positive responses in patients with high-risk characteristics, such as those with a high Ki-67 proliferation index. Taken together, these results further underline the benefits of KTE-X19 and support further investigations into this treatment protocol.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
In your experience, what is the average vein-to-vein time when treating patients with DLBCL with a reimbursed CAR T-cell therapy (from apheresis to infusion)?


